“A Sanger sequencing gel carries many DNA bands for each reaction and is thus complicated to study. In this article, Learn how to interpret a Sanger Sequencing gel with thumb rules.”
Do you know the Human Genome Project was conducted in different laboratories worldwide and sequenced 3.2 billion DNA bases? HGP also identified around 23,000 genes as well.
The Sanger sequencing technique was developed by Sanger and co-workers in 1977. It is a chain termination method. The principle of Sanger sequencing demonstrates that during DNA synthesis, the addition of ddNTP instead of dNTP terminates the synthesis.
When all four ddNTPs are included in the reaction, they cause the synthesis process to stop whenever they come across their complementary nucleotide in the DNA chain— this termination of synthesis results in a single fluorescence signal, which is then recorded.
However, traditionally, all four separate reactions were run on a gel, fragments were separated based on their size and used to interpret the results. Techniques like capillary electrophoresis replaced the conventional method with automation and high-end precision.
However, despite advancements in sequencing technologies, certain laboratories continue to rely on the traditional method of Sanger sequencing, which remains effective. In fact, it was Sanger himself who pioneered the use of gel electrophoresis to analyze sequencing results.
To enhance your understanding of sequencing techniques, this article will provide a comprehensive explanation of the sequencing gel and guide you through the process of interpreting Sanger sequencing gel results.
Stay tuned.
Key Topics:
Sanger sequencing gel:
A Sanger sequencing gel is a Polyacrylamide gel and known as PAGE— polyacrylamide gel electrophoresis. The porous medium prepared by the gel matrix helps DNA fragments to migrate and separate.
The principle of PAGE is similar to the agarose gel electrophoresis. Under the influence of electrical current, DNA fragments can migrate in a gel. Based on their size, they run at different speeds and separate.
To prepare a Sanger sequencing gel, polyacrylamide gel solution is poured into the gel caster with a gel comb. A gel comb creates wells to load the sample. The gel is allowed to solidify and the comb is removed, afterward.
The gel is placed in the pre-buffer-filled gel tank, carefully until it is completely submerged in the buffer. Four separate sequencing reactions are mixed with the loading dye and loaded in individual gel well.
The sample is allowed to run for an hour or two (approximately) to separate every possible DNA fragment in the gel. After run completion or the loading dye reaches to adequate distance, the current is turned off and the gel is sent for interpretation.
The gel is placed in the UV light source to read the results. An expert takes a photograph or copy of the results and conducts an analysis part.
How to read Sanger Sequencing results?
Suppose you have an assignment, your teacher wants you to learn how to read Sanger sequencing gel and you don’t have any idea how to start. Closely understand this part of the article to learn it.
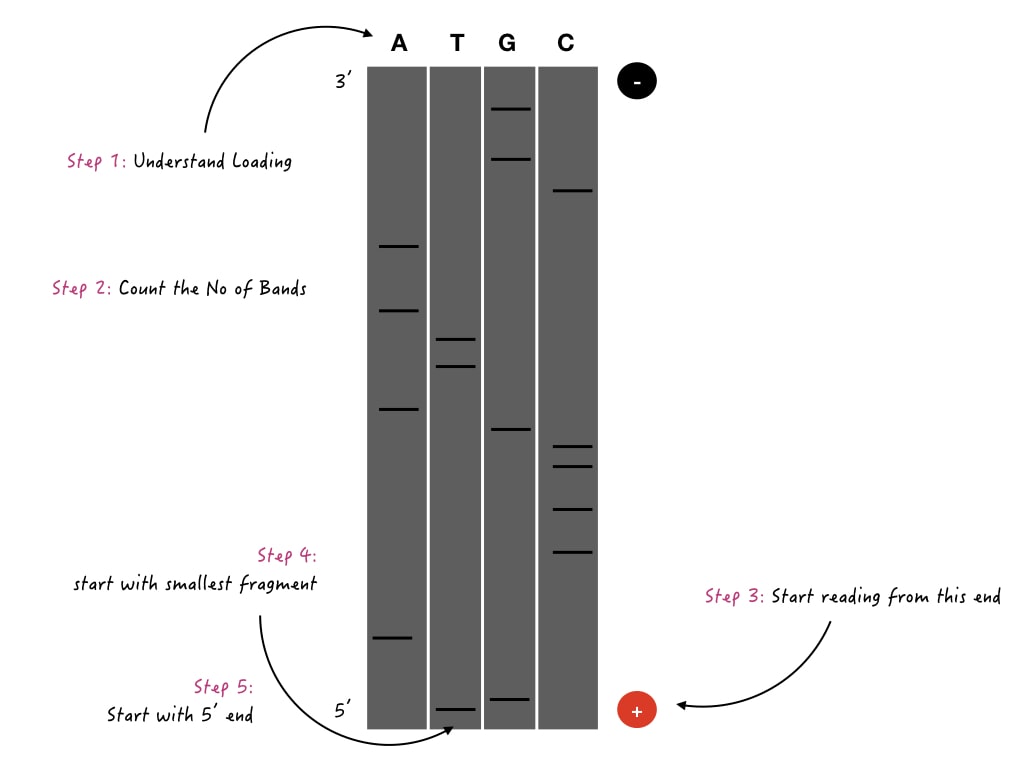
Let’s start.
Step 1: Understand the loading
Label your picture. Or examine in which well you have loaded which ddNTP sample. You possibly have four lanes for each chain termination reaction. Closely look at the image I have provided.
You have loaded in this sequence A, T, G and C. So in lane 1, we have loaded a sample for ddATP, in lane 2 for ddTTP, lane 3 for ddGTP and lane 4 for ddCTP.
Step 2: Count the bands
In the very next step, count the number of bands you will observe in each lane individually and in total. This will help you to cross-verify the results. In our example, the number of bands in each reaction is given here.
| A | T | G | C | Total |
| 4 | 3 | 4 | 5 | 16 |
So when we finally write down our sequence, it must have a total of 16 nucleotides and 4, 3, 4, and 5 nucleotides for A, T, G and C, respectively.
Step: 3: Star with the positive
All the samples would run under the influence of current and as DNA is negatively charged, it migrates from the negative to the positive side. We have to start reading from the positive side of the gel (Thumb rune).
Step 4: Start with the smaller sized-fragment
Smaller fragments run faster and vice versa. So fragments away from the gel wells are smaller in size, while the fragments nearer to the gel wells are larger in size. In other words, the fragments more on the positive side are smaller in size and vice versa (Thumb rule).
You have to start sequence decoding from smaller-sized- fragments, meaning from the positive node side.
Step 5: start writing from 5’
The DNA sequence should always be written from the 5’ end. If you don’t know how to write a DNA sequence and its complementary, read this article. DNA synthesis always occurs from a 5’ to 3’ direction.
Meaning, the polymerase starts adding nucleotides from the 3’ end on the opposite strand, and thus at the end leaves a free 5’ end. So the sequence should be started writing from the 5’ end.
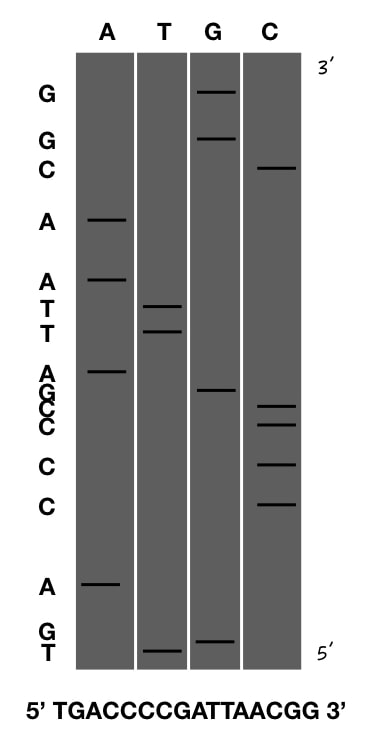
Step 6: Start writing the sequencing
Now combine all four rules, re-read them, observe the image and start writing the sequence. Three, two and one! Go. My sequence is here: TGACCCCGATTAACGG.
Wrapping up:
This is just a practice example with a few base pairs, but in an actual Sanger sequencing gel, there could be hundreds to thousands of base pairs. It’s important to note that the quality of the DNA bands may vary, posing challenges in interpreting the gel. Therefore, it’s crucial to have an expert cross-verify any sequencing results before reporting.
We hope you enjoyed this article! Don’t forget to share it and bookmark the page. If you’re interested, you can explore more articles on this topic in our dedicated category: DNA sequencing.