“An organic substance present in the nucleus of a living cell that stores and transfers information is known as the nucleic acid.”
DNA and RNA are the two types of nucleic acid present in all organisms (RNA is some viruses). Although the term “nucleic acid” was coined by Richard Altmann in 1889, it was actually discovered by Friedrich Miescher in 1869. He named it “nuclein”.
The nucleic acid in the form of either DNA or RNA is present in all cells in all organisms on the planet earth, no one can live without it.
The actual structure of one of the nucleic acid- DNA was determined by Watson and Crick in 1953. We have tons or articles on the present topic, and so ain’t discuss everything in detail. You can read it on our blog.
Both types of nucleic acids are known to science students, however, usually, they don’t know the difference between monomer, dimer and polymer.
In the present article, I will explain different forms of nucleic acid, its importance and other related information. Note that we will discuss the structure of it in general. I hope this article will encourage you to learn more about genetics.
Stay tuned,
Key Topics:
Structure of nucleic acid
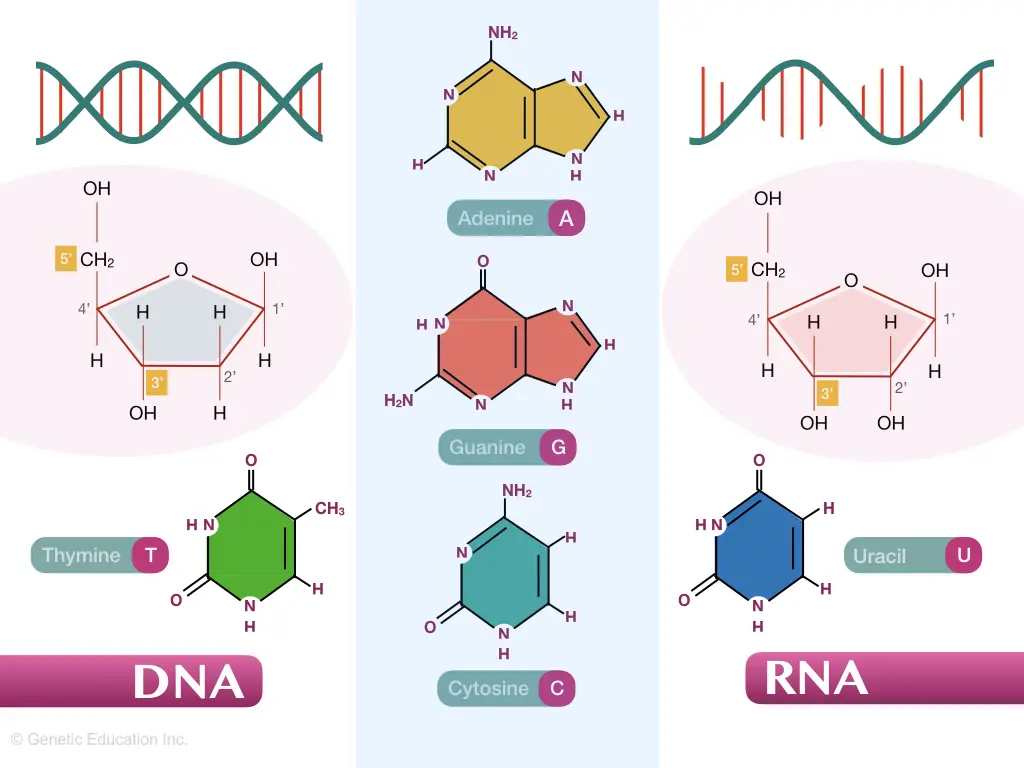
In general, either DNA or RNA- nucleic acids are made up of three major components- sugar, phosphate, and nitrogenous bases.
Purines and pyrimidines are present as nitrogenous bases in DNA while ribose and deoxyribose as sugar are present in RNA and DNA respectively. DNA and RNA have substantial differences in terms of structure and function, to know more you can read this article:
Nucleic acid is a complex molecule containing a long chain that consists of millions of monomer units, we can say that it’s a biopolymer. Commonly, students may not know the differences between the monomer and polymer, don’t worry I will cover it in the upcoming section.
The nucleic acid is a long chain of nucleotides and we know it as a polynucleotide chain. It encodes a protein. The polynucleotide chain indeed is made up of many monomer units of nucleotides
The monomer of nucleic acid:
A single unit of the whole polynucleotide chain which is the nucleotide is known as the monomer.
Mono- “one” + mer- “part”
Now you may wonder that nucleic acid is made up of sugar, phosphate and nitrogenous bases and what’s a nucleotide? Let me explain to you that a sugar, phosphate and nitrogen base collectively manufacture a structure known as “nucleotide”.
Notedly, when phosphate isn’t present, it is known as a nucleoside. This article will clarify things precisely.
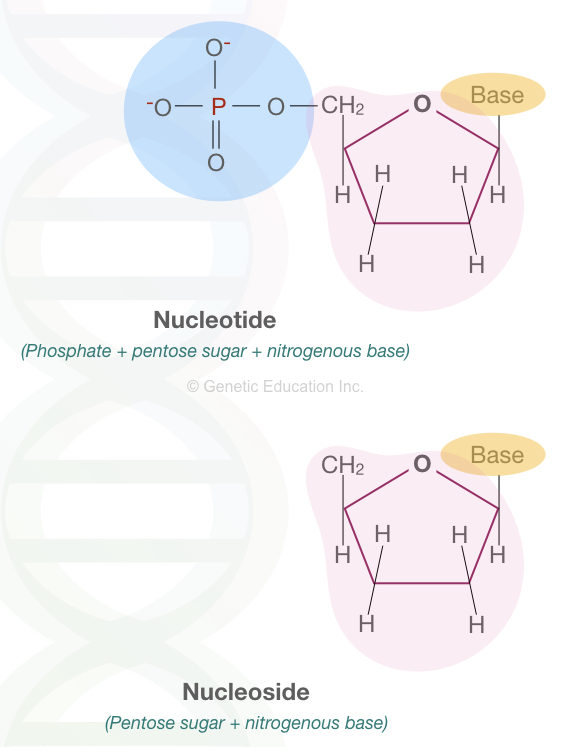
Remember:
Base and nucleotides are different. When we call base pairing, it simply means one nitrogenous base binds to another complementary nitrogenous base on another strand by hydrogen bonds.
But when we call nucleotide, it is a unit of sugar + base + phosphate that binds to an adjacent nucleotide with the phosphodiester bond. See the image below,

Interestingly, some nucleic acid monomers are also found on the surface of meteorites. Scientists believe that the nucleic acid polymer is formed from this monomer by natural events and that might be a reason for the evolution of life on earth.
However, less scientific evidence supports this fact.
Dimer of nucleic acid:
When a monomer of nucleic acid- nucleotide binds with the adjacent nucleotide with a phosphodiester bond it is called a dimer of nucleic acid or dinucleotide.
Similarly, when three nucleotides join together by the phosphodiester bond it is known as trinucleotides. Tones of triplet repeat units are present in our genome; in coding and non-coding regions. When it grows abnormally it causes serious genetic problems. For example, Huntington’s disease comprises abnormal expansion of CAG triplet nucleotide repeats.
Dinucleotide repeats are also found in a genome- a type of tandem repeats responsible for hereditary nonpolyposis colon cancer is a dinucleotide repeat.
NAD (Nicotinamide adenine dinucleotide)- a type of dinucleotide functions as a cofactor in metabolic reactions. Two nucleotides adenine and nicotinamide join together by a phosphodiester bond and form NAD.
The polymer of nucleic acid:
A long chain of nucleotide monomers is referred to as a polynucleotide chain (a polymer of nucleic acid).
Poly- “many” + mer- “part”.
Our genes are actually long polymers and manufacture various proteins, however, non-coding DNA is also made up of polymer units.
The function of nucleic acid:
The key function of nucleic acid is to transfer information, more precisely, genetic information from one to another cell, consequently from one to another generation.
Our cell is like a computer having nucleic acid as a storage unit; utilized for storing and transferring information just like the hard disk of the computer. Every single monomer of nucleotide certainly is a storage unit (just like every byte of computer memory).
The computer program works on the binary language of 0 and 1 while our nucleic acid- DNA stores information in A, T, G and C. Three of the four nucleotides form a codon, specifically called a “triplet codon”.
Each triplet codon forms a monomer of amino acids. When each triplet codon forms each amino acid, it eventually makes a protein. The mechanism works as a central dogma processing of the cell.
Although storing and transferring information is the main function of nucleic acid, replication, transcription, and translation is a way to do this.
By replication, the DNA (one of the types of nucleic acid) copies the information, creates exactly the same polynucleotide chain and transmits it to the daughter cell.
Transcription makes an mRNA from the DNA which truly consists of the coding information. Finally, translation manufactures a mature protein from the mRNA.
Yet another key function is to protect the genetic code from enzymatic lysis.
Interestingly, our energy sources Adenosine triphosphate (ATP) and guanosine triphosphate (GTP) are triple monomer units, a life required as an energy source for performing various metabolic activities. GTP also has a significant role in G-protein mediated cell signaling.
Genetic code:
It’s also essential to introduce the genetic code in the present chapter for understanding the chapter precisely.
The mRNA having coding information, processed at ribosome at were each three bundles of nucleotides- “triplet”, read as one codon for making an amino acid. Keep in mind that the genetic code consists of triplets of nucleotides, not nitrogen bases.
Each genetic code together makes a polypeptide chain and finally a protein. Processing removes non-coding introns and collects only exons. It’s just an introduction for the topic, to know more you can read this article:
Properties of nucleic acid:
- It is acidic in nature, accepts paired electrons and donates protons.
- The nucleic acid is overall negatively charged due to the presence of a negative charge on the backbone.
- It’s colorless and odorless.
- Nucleic acid is insoluble in alcohol so it can precipitate. Other chemicals that precipitate nucleic acid are HCl and acetic acid, although less preferred
- It dissolves easily in hot water.
- A nucleic acid DNA is a double-stranded and antiparallel molecule so DNA is more stable than RNA.
Synthetic nucleic acid and nucleotides:
Synthetic nucleic acid, nucleotides and polynucleotide chains have varieties of applications in genetics ranging from gene therapy to gene transfer. A specific/target/faulty DNA region or gene can be replaced by a new synthetic gene or polynucleotide chain.
Yet another common example of synthetic and artificially synthesized nucleic acid is the PCR primers, which scientists use for copying DNA, in vitro.
Also, assays like the Polymerase Chain Reaction and DNA sequencing need dNTPs and ddNTPs, respectively, for synthesizing the DNA or target and identifying the sequence.
Threose nucleic acid, locked nucleic acid, glycol nucleic acid and peptide-bound nucleic acid are several types of synthetic nucleic acid having definite applications in various biological research fields.
Conclusion:
So that’s it for this topic, I hope this article will clarify basic fundamentals of genetics especially, regarding the monomer, dimer and polymer of nucleic acid.
Every organism which possesses DNA as nucleic acid has an almost unchanged nucleic acid structure, though may have sequence variations. One fascinating study suggests that RNA evolved far earlier than DNA. DNA ultimately forms RNA, then why DNA was evolved is still a mystery.
Resources:
- Real D. (2016) Physical and Chemical Properties of Nucleic Acids. In: Micic M. (eds) Sample Preparation Techniques for Soil, Plant, and Animal Samples. Springer Protocols Handbooks. Humana Press, New York, NY.


