“Plant species are heterogeneous, contain a differential cell composition and cell wall structure and thus, possibly need a separate DNA extraction protocol for every species.”
Plant DNA extraction is a difficult process, let’s find out why.
Every living organism on earth has genetic material as either DNA or RNA, however, prominent is DNA. Plants, like mammals, contain genomic DNA in their cell nucleus and organelle DNA in cytoplasm, for instance, plastid or chloroplast DNA in chlorophyll.
It is important to know that plants (and mammals) don’t have genetic material in the form of RNA. Excluding only a few viruses (which are even not considered living entities) every organism from prokaryotes to eukaryotes comprises only DNA.
Plants are powerhouse because they produce food by using their unique cell organelle- chlorophyll. We lack chlorophyll in our cells. And that’s why we can’t synthesize our own food and we need plants to survive on earth.
Save plants and plant more trees!
Anyway, coming to our main point.
Isolation of plants’ DNA allows scientists to understand how various plant systems work and how and why they are different. Recent advancements in genetic studies and genetic engineering help scientists investigate and alter a specific plant phenotype.
Scientists use a technique called recombinant DNA technology to introduce a new gene in a plant cell to alter the gene and thereby produce economically important plant species. For example, the successful alteration and production of Flavr Savr tomato by antisense RNA technique.
However, for all such research, we need DNA, a pure and good amount of plant DNA. The heterogeneous nature of plants and differential cell composition make plant DNA extraction a tedious, difficult, time-consuming and costly process. You are on this article, which means you know it is a difficult intervention.
So we need separate protocols or modifications for different plant species. And after doing many such optimizations, we would not get a good extract. The reason is first we require to understand why plant DNA extraction is a difficult process.
In this article, I will explain why it is difficult to extract DNA from plants and possible optimizations to overcome it.
Stay tuned.
Key Topics:
Why is plant DNA extraction difficult?
Three structural entities of a plant cell viz polysaccharides (pectin, cellulose, chitin and amylopectin), secondary metabolites (polyphenolics, tannins, flavonoids, lignins etc) and proteins and non-nuclear DNA hamper the plant DNA extraction
It, eventually, affects the quality and quantity of extract. The concentration of each of these components, tissue type, plant part and age of tissue are some factors that affect the overall DNA extraction process, negatively.
As the concentration of these ingredients varies from plant to plant, a separate DNA extraction protocol is required for each. These are usually common contaminants of plant DNA. I will explain each one separately here to give more clarity.
Polysaccharides:
Polysaccharides have a prominent structural role in the plant cell wall. It is present in the form of cellulose, pectin, chitin or xylans, amylopectin etc. Functionally, it provides stability to plant cells. It creates a hard, rigid and tight cell wall structure in higher plants.
Such a strong structure helps to generate turgor pressure. Almost all plants contain any type of polysaccharide as a constituent of the plant cell wall.
On the technical side, polysaccharides can co-precipitate with DNA and give us a false indication of higher DNA yield. However, a careful examination shows a sticky and viscous appearance revealing contamination of polysaccharides.
Furthermore, polysaccharides prevent catalytic activities of enzymes like ligase, polymerase, and endonuclease, henceforth it is important to remove them before using DNA in PCR, recombinant DNA technology and restriction digestion.
Polyphenols:
Aromatic or medicinal plants have huge amounts of secondary metabolites in the form of polyphenols. And generally, we are very much interested in such plant studies to know their medicinal importance.
Unfortunately, extracting DNA from these groups of plants is burdensome.
Flavonoids, tannins and terpenoids, lignin, carotenoids, etc are common polyphenolic compounds present in the plant cell granules. They are one of the most common contaminants that exhibit a significantly adverse effect on DNA extraction yield and quality.
It remains attached to nucleic acid once it comes out from granules. Different plants have different groups and concentrations of polyphenols and are unlikely to encounter using a single protocol.
Polyphenols are a good source of antioxidants for us and they should be in our daily diet.
Higher amount of protein and RNA:
Plants also have a higher amount of various proteins. In addition, in the extraction process, the RNA often creates problems too. Overall, protein and RNA are the other two contaminants with which we have to deal.
These two contaminants, however, can be easily removed by using chemicals and enzymes. Proteins and RNA may sometimes co-precipitate with the DNA and even give bands in a gel. Careful washing is also extremely useful to get the high-quality extract.
Non-nuclear DNA:
Plants contain a higher amount of non-nuclear/ organelle DNA in mitochondria and chloroplast. Often known as cytoplasmic DNA, non-nuclear DNA greatly affects several downstream processes and constitutes problems in genomic DNA library preparation.
In mammals, it isn’t a great deal, but in plants, we should have to care about it. Anyway, we have a solution for that as well.
Solutions to overcome these problems:
I spent days and nights in my genetic lab during my masters and extracted many plant DNA, although I have expertise in medical genetics, I can handle plant samples too. I and my colleagues have worked on various plant species like Bauhinia Variegata, Datura stramonium and Ocimum tenuiflorum etc and got good results as well.
So I have a decent working hand on plant DNA extraction. We have highly optimized protocols to obtain a great yield. Here are some how to overcome ‘tips’ from my side. And trust me it will work.
How to remove Polysaccharide contaminants?
To deal with the polysaccharides use a higher salt concentration solution, for example, 1 to 6mM NaCl in the extraction buffer.
Use CTAB, which helps prevent nucleic acid and removes the protein part and polysaccharide through the insoluble complex.
Also, you can use the pectinase, and cellulase enzyme in the extraction protocol.
Tip to success:
Treat the sample twice or thrice with the extraction buffer to get excellent results.
How to remove Polyphenolic contaminants?
The oxidized form of polyphenols converted by the oxidase enzyme remains bound to the DNA. The use of antioxidants can help to overcome this problem.
PVP (Polyvinyl pyrrolidone) and PVPP (Polyvinyl polypyrrolidone) are two common antioxidants that can work finely with your plant DNA extraction protocol. Do not forget to use it. However, keep in mind to maintain a low pH environment to improve the effect.
If the problem still persists, use other agents like beta-mercaptoethanol, ascorbic acid, DTT, sodium sulfate, sodium azide and sodium iso-ascorbate in the extraction.
How to remove proteins and RNA contaminants?
CTAB, SDS and beta-mercaptoethanol are three common ingredients of the plant cell lysis buffer and are sufficient for cell lysis. They can remove proteins easily by distorting their structure.
An enzymatic approach using the proteinase K can also work effectively. Use the appropriate amount of proteinase K, as instructed by the manufacturer with adequate incubation conditions.
Moreover, RNase can be used to remove RNA from the extraction.
How to remove non-nuclear DNA?
See technically, one can part the process into cell wall lysis and membrane lysis. As the non-nuclear DNA is present in the cytoplasm, through cell wall disruption, cytoplasmic impurities/contaminants can be removed, ideally.
Triton X 100 stabilizes the nuclei while other ingredients of lysis buffer disrupt the cell wall. In addition, the use of non-ionic detergent or nuclear membrane stabilizer in the tissue homogenization process improves the overall genomic DNA yield.
Summary:
| Chemical | Concentration | Function |
| CTAB | 2 to 5% (v/w) | Removes proteins |
| SDS | 2% (v/w) | Removes proteins |
| Beta-mercaptoethanol | 0.3 to 2% (depending upon the polyphenolic content) | Inactivates and removes polyphenolic compounds. |
| Sodium chloride | 1 to 5M | Removes polysaccharides |
| Triton X 100 | 0.5 to 0.8% | Removes chloroplast and mitochondrial DNA. Stabilize nuclei |
| PVP | 1% | To remove polyphenolics |
Note that in the buffer, other ingredients like Tris and EDTA are also required. Refer to your lysis preparation guide or standard protocol, use these ingredients if required.
Bonus tips:
How to improve plant DNA extraction?
First thing is to read. Read about the plant species from which you want to extract DNA, and understand its cell and cell wall composition, polysaccharide structure and polyphenolics it produces.
This will give you a theoretical brief idea about what optimization you need to do. For example, if you are working on an aromatic plant with a very high amount of secondary metabolites, increasing the concentration of NaCl, PVP or PVPP and beta-mercaptoethanol would work.
Trust me! your 70% work will be completed only by reading.
Use fresh tissue samples.
Younger leaves contain less starch and secondary metabolites, and a large number of dividing cells, thus use a young, healthy and tender plant leaf as a sample. Avoid using older and dried tissue samples.
Use enzymes like proteinase K, RNase and cellulase in the extraction process, if you repeatedly fail. Enzymes work more effectively than chemicals.
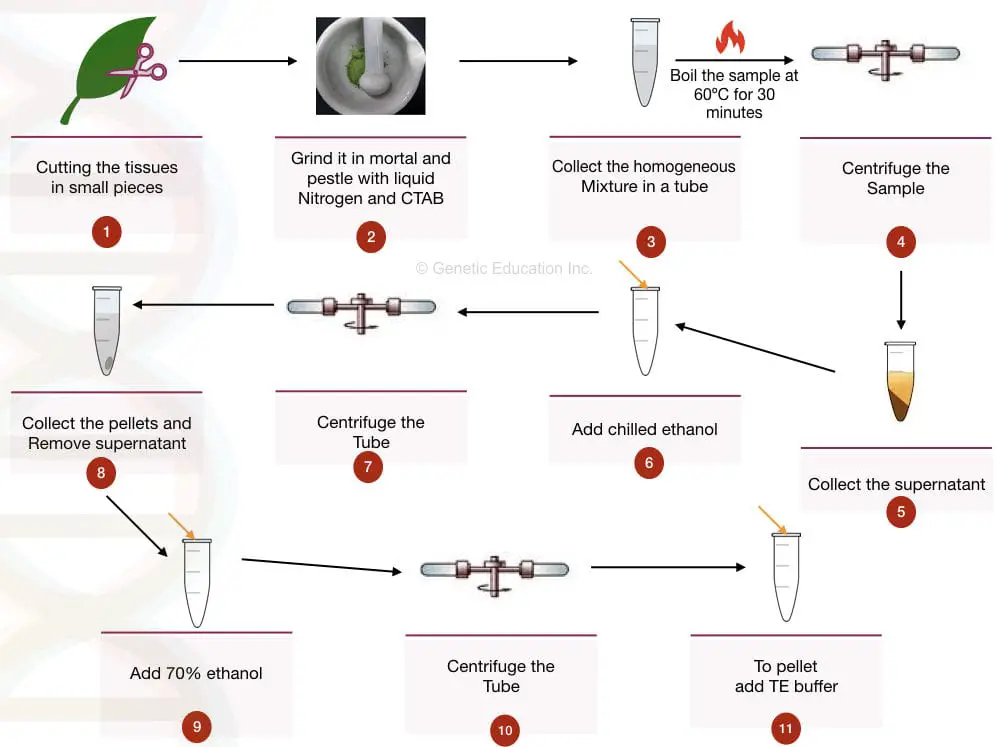
Tissue homogenization is a very important part of plant DNA extraction. Use liquid nitrogen and grind tissue well and vigorously. Try to make fine powder.
Store precipitate overnight and re-precipitate after 24 hours, this will surely improve the yield of DNA precipitates.
Make your observation power stronger. Learn from previous failures. For example,
If you get sticky and oily precipitate- repeat the process.
If you get a greenish precipitate- repeat the process
If you get precipitate without a white color- repeat the process
If you don’t get precipitated- repeat the process.
Such physical examinations are indications that the extraction isn’t great. The DNA precipitate would be whitish- cotton-like threads.
Make a log book and must note down the alteration you did. It will help you in optimizing the process and helps in future extraction.
Use an excellent lysis buffer for plants. Here is the recipe.
- NaCl – 2M
- CTAB – 3%
- PVP – 1%
- Tris HCl – 100 mM
- EDTA – 25 mM
- Beta-Mercaptoethanol- 0.2%
Read more: CTAB DNA extraction buffer for plant DNA extraction
Wrapping up:
Pure and high-yield DNA is a prerequisite for any genetic experiment. Contaminated DNA may cause faults and problems in further processing and downstream analysis, especially for plant DNA.
To use the DNA or protocol directly in serious experiments like sequencing or recombinant DNA, and standardize it for the plant species you are working with, results will mislead, otherwise.
Plant DNA extraction isn’t rocket science to optimize, you have to keep two things in mind, one size can’t suit all and master the skills to use different ingredients.
You require to develop problem-solving skills and how to use different chemicals and compositions to overcome that.