“Microsatellite instability is defined as indel (insertion or deletion) mutations within the repetitive regions (microsatellite) in cancer cells by the inactivation of DNA mismatch repair proteins.“
Do you know?
- Introns usually comprise microsatellites.
- 3% of the human genome contains microsatellites.
- It is a non-coding region present on the telomere.
- Such regions are repetitive in nature and used for DNA tests.
Cancer occurs through various genetic mechanisms, starting from a single base insertion or deletion mutations to complex copy number variation or loss of heterozygosity.
Cancer is attributed to not only genetics but also to other factors and so is a complex process. There are many biological markers scientists use to screen a particular type of cancer or group of cancers.
Biological markers:
Any quantitative alteration in biological molecules- DNA, RNA or protein which is present in cancer is defined as a biological marker for cancer.
Microsatellite instability short for MSI is one such biomarker for Lynch syndrome and colon cancer. It occurs when the repeat regions of a microsatellite abnormally grow by any hypermutation phenomenon.
Such hypermutation occurs by the error in replication and is attributed to the failure of DNA mismatch repair proteins. But as you know that microsatellites are non-coding and used in biological verification (such as DNA tests), how does it cause disease? why do repeat numbers change and how the instability is associated with cancer?
These and all other questions we will answer in the present article. The topic is a bit complicated, I will try my level best to make you understand in layman.
Stay tuned
Key Topics:
What is microsatellite instability?
By definition, the microsatellites are smaller, with 2 to 6 nucleotides long, repetitive and non-coding regions. The regions are usually located on telomeres and between the coding sequences of a gene and are highly polymorphic among individuals.
No two individuals have a similar number of repeats, however, at the same time, it remains stable as well. Replication, when occurring accurately, at a normal pace results in intact and accurate microsatellite synthesis.
Often time, replication fails to properly synthesize microsatellites. Two reasons for that are first, most of the microsatellite sequences are located on extreme ends and second, polymerase sometimes loos its activity before reaching the ends.
However, the backup mechanism, known as DNA mismatch repair eventually fills the gap remained by replication errors and completes the process. It makes microsatellites intact.
But oftentimes, dMMR (DNA mismatch repair) proteins can’t complete the job, upon either inactivation or loss of function. These also occur by a mutation in genes coding for such proteins. And in consequence, it results in microsatellite instability.
The repetitive nature of SSR markers (SSR are our microsatellites) makes them more prone to mismatch thereby hypersensitive towards mutation. As the length increases the probability of getting mutation increases as well.
This clarifies that larger microsatellite repeats are at higher risk of getting instability. This is the simplest explanation of the process. Now let us understand MSI more elaboratively and scientifically.
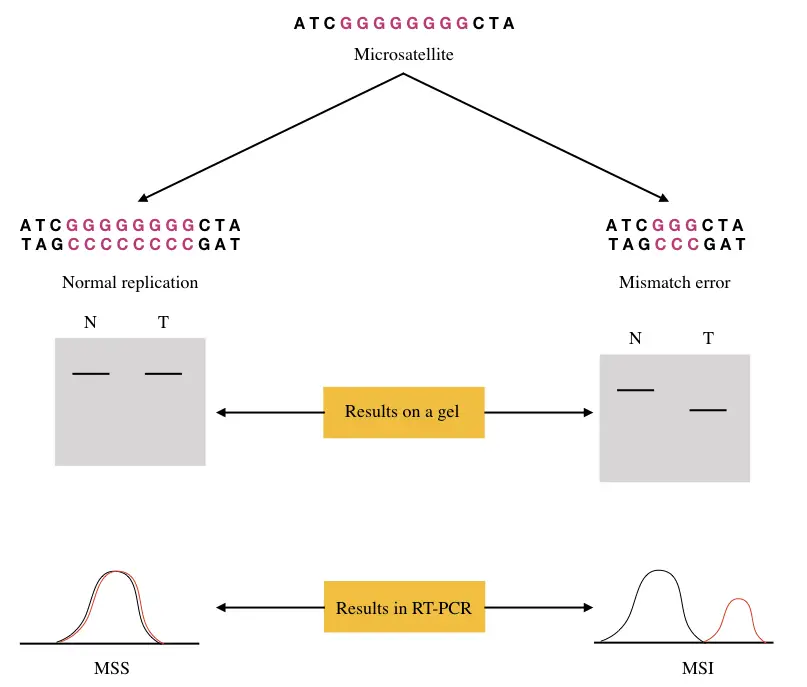
Mechanism of MSI:
Replication occurs at the S phase of cell division. It does perform DNA synthesis and DNA proofreading (process to correct replication errors) and constructs a new DNA strand and repairs the faulty one. However, studies suggest that the replication efficiency decreases upon reaching the ends.
Often known as the replication problem’ when occurs, the polymerase can not synthesize and/or proofread the DNA accurately, especially at the ends, viz at microsatellite regions. As mentioned, the repetitive nature of microsatellites makes it harder for the polymerase to do the job correctly and results in mismatches.
So a specific cell mechanism, known as DNA mismatch repair, as a backup repairs mismatches especially when replication errors occur. MLH1, MSH2, MSH6 and PMS2 are four genes that encode proteins mutL homologue 1, mutS homologue 2, mutS homologue protein 6 and postmeiotic segregation increased 2, respectively. These four proteins in synchronization perform DNA mismatch repair at microsatellite regions.
Meaning it repairs each and every mismatch possibly to regulate the integrity of every microsatellite. However, by somatic or germline mutations the inactivation of either gene results in loss of function protein causing dMMR failure.
In cancerous cells by any means when this emerges, results in hypermutation of various microsatellites. So cancer with microsatellite instability has thousands of mutations or more in such regions. Several mechanisms of MSI is explained here:
Replication strand slipped and mispairing: During replication, especially during tandem repeat synthesis the newly formed allelic strand mismatches with the parental one and leads to MSI.
dMMR protein deficiency: The DNA mismatch repair system including dMMR proteins is capable enough to repair slipped mispairing, however, inactivation of any protein or all proteins of the dMMR pathway fails to repair the slipped strand.
Such loss of function mutation may occur by a mutation or epigenetic alterations in the dMMR protein-coding genes.
Deletion/insertion within MS: often time deletion or insertion of nucleotides within any of the MS results in microsatellite instability. Such ‘indels’ occur either during the process of replication or randomly under the influence of external stimuli.
Conclusively, MSI results in abnormal gain or loss of mono, di or trinucleotide repeats of genomic microsatellites at a higher rate than in normal cells.
Two mononucleotide and three dinucleotide microsatellites, commonly found in cancer are BAT25 & BAT26 and D2S123, D5346 & D17S250, respectively. Notedly, depending upon how many SSR are hypermutated, the condition can be categorized into three broad categories viz MSI-H, MSI-L and MSS.
MSI-H
The microsatellite instability-high occurs when instability is reported in more than two of the 5 common SSR. Such a condition is commonly reported in many types of cancers including colon cancer and Lynch syndrome.
MSI-L
The microsatellite instability-low occurs when instability is reported in a single SSR marker. MSI-L is not so prevalent in cancer, although it shows the likelihood of getting cancer.
MSS
The microsatellite stable is when none of the markers show instability. MSI-L and MSS are considered similar as are not reported in cancer, however, as I mentioned, MSI-L increases the genetic predisposition to cancer.
MSI and cancer:
Altogether, the instability of microsatellites (SSR markers) results in both germline and somatic cancer. Lynch syndrome and colon cancer are classic examples of germline and somatic cancer by MSI, respectively.
MSI in Lynch syndrome:
Lynch syndrome is an autosomal dominant condition that occurs by mutation(s) in DNA mismatch repair genes during germline development, Henceforth is a type of inherited cancer.
1 out of 279 people have Lynch syndrome and the majority of them don’t know about it.
-Promega Corporation (Promega is a leader in microsatellite instability research).
Mutation in MLS1, MSH2 or MSH6 causes Lynch syndrome condition. It increases the chances of the likelihood of getting various types of cancers. Colon, colorectal, liver, gastritis, gallbladder, pancreatic, bone, ovarian, uterine, cervical and vulvar cancer are the most common cancers in Lynch syndrome.
Although the condition is inherited, the tumor doesn’t start developing by birth, instead, tumor progression appears between the age of 20 or 30 and the individual may have life long risk of developing cancer.
Depending upon the alteration of the gene, the Lynch syndrome can be classified into four categories of MSH2, MSH6, MLH1 and PMS2 in which MLH3 and exo1 are candidate genes involved in the development of tumors by Linch syndrome.
Hereditary non-polyposis colorectal cancer (HNPCC) is another common name for Lynch syndrome that causes hereditary colorectal cancer.
However, it is notable that MSI test-positive for Lynch syndrome doesn’t mean to have cancer or likely to have it. The family history of the individual is taken into consideration before predicting cancer.
Colorectal cancer:
It is a type of sporadic colon cancer, majorly, but is also inherited in a small number of cases. The sporadic nature of colorectal cancer is attributed to genetic polymorphism, epigenetic alterations and environmental changes.
It is occur by the sporadic mutation in the dMMR protein-coding genes which eventually fails to stabilize microsatellites. MSI-PCR-based detection methods are used for the detection, diagnosis and prognosis of colon cancer.
MSI in other tumors:
| Tumor | % of MSI | Note |
| Colorectal cancer | 15% | |
| Lung cancer | 0 to 40% | |
| Gastric cancer | 15% | |
| Endometrial cancer | 33% | Defective MRR |
| Cervical cancer | 5% | |
| AVC (Ampulla of Vater Cancer) | 10% | |
| Breast cancer | 1% | |
| Esophageal cancer | 5% | |
| Ovarian cancer | 10% | 1% cases have germline MRR mutation. |
| Head and neck cancer | 1% | |
| Prostate cancer | 1 to 12% | Mononucleotide markers |
These data suggest that MSI has been involved in the development of many types of the tumor by germline or sporadic mutations. And therefore MSI- testing becomes very critical in such cases.
MSI testing:
Among available methods for the detection of MSI, molecular methods such as PCR or DNA sequencing are more accurate and reliable. Here in this section, we will discuss some of the common methods used for the detection of MSI.
Multiplex PCR:
Multiplex PCR using fluorescence and/or capillary electrophoresis is the gold-standard method for instability detection. It provides reliable results with high specificity and sensitivity and nearly 100% accuracy.
A single multiplex reaction can amplify and detect all five common microsatellite markers (BAT-25, BAT-26, D2S123, D5S346 and D17S250). The first two are single nucleotide repeats and the rest of the three are multi-nucleotide repeat sequences.
Fluorescence-based PCR assay is powerful enough to detect MSI- H, MSI- L and MSS viz, polymorphism in more than two markers, in a single marker and in none of the markers, respectively. Moreover, the use of capillary electrophoresis increases the efficiency of detection many folds.
Next-generation sequencing:
Yet another accurate molecular testing option for MSI detection is NGS. It sequences all the possible common and uncommon microsatellites and instability (if any) by rapid and robust sequencing technology.
The NGS is most accurate, but at the same time is a costly and time-consuming process. Further to this, it can test many genes and alterations associated with tumors.
Immunohistochemistry (IHC):
The IHC method is a simple, effective and rapid method for the indirect detection of MSI. It indeed measures the expression of dMMR (DNA mismatch repair) protein using quantitative analysis. Under expression of one or all four proteins indicates MSI while adequate expression of all proteins indicates stable MS.
Noteworthy, the technique is not as accurate, sensitive and specific as the PCR.
Wrapping up:
Microsatellite instability occurs by a defective DNA mismatch repair pathway. A mutation in genes MSH2, MSH6, MLH1 and PMS2 may cause the failure of the dMMR system. However, oftentimes it remains intact.
MSI-H, MSI-L and MSS are three states of instability among which MSI-H is considered a high-risk group for cancer. It doesn’t warranty cancer, though.
Microsatellite instability testing is an important tool for tumor study and cancer detection and prognosis. The multiplex PCR assay is by far the most accurate and gold standard detection technique.
Sources:
- Li, K., Luo, H., Huang, L. et al. Microsatellite instability: a review of what the oncologist should know. Cancer Cell Int 20, 16 (2020). https://doi.org/10.1186/s12935-019-1091-8.
- Boland CR, Goel A. Microsatellite instability in colorectal cancer. Gastroenterology. 2010 Jun;138(6):2073-2087.e3. doi: 10.1053/j.gastro.2009.12.064. PMID: 20420947; PMCID: PMC3037515.
- Nojadeh JN, Behrouz Sharif S, Sakhinia E. Microsatellite instability in colorectal cancer. EXCLI J. 2018 Jan 22;17:159-168. doi: 10.17179/excli2017-948. PMID: 29743854; PMCID: PMC5938532.
- Oncology pro- Microsatellite instability- defective DNA mismatch repair: Biomarker factsheet.
- Lu Y, Soong TD, Elemento O (2013) A Novel Approach for Characterizing Microsatellite Instability in Cancer Cells. PLoS ONE 8(5): e63056. https://doi.org/10.1371/journal.pone.0063056.
- CDC, hereditary colorectal cancer.


