“An increased tendency of the genome to acquire mutations is known as Genetic instability. Explore the concept, definition, types, mechanisms, and detection methods of genetic instability in this article.“
Genetic stability is one of the hallmarks of cancer, in which our genome becomes unstable and causes cancer. Cancer is genetic, meaning, genetic mutations lead to cancer.
Change in the DNA or nucleotide sequence, or chromosome structure or numbers often reported in various types of neoplasia. Interestingly, genetic instability is a condition in which the genome’s tendency to acquire mutations increases enormously.
In this article on our cancer genetics series, I will explain to you the concept of genetic instability, its definition, types, mechanisms and detection methods.
Stay tuned.
Disclaimer: The content presented herein has been compiled from reputable, peer-reviewed sources and is presented in an easy to understand manner for better comprehension. A complete list of sources is provided after the article for reference.
Key Topics:
What is Genetic Instability?
The term “genetic instability” describes a range of DNA or chromosomal alterations that permanently affect genetic information, from point mutations, deletions, and insertions to chromosomal rearrangements and, in some cases, chromosome number changes.
In simple words, the genome becomes less stable and starts acquiring various mutations, randomly.
A person with genomic instability will have an increased rate of mutations (deletions, insertions, inversions, etc.), structural chromosomal abnormalities or rearrangements, and numerical chromosomal aberrations.
In normal conditions, our genome maintains high fidelity by various mechanisms such as DNA repair pathways, cell cycle checkpoints, and apoptosis. But in conditions such as cancer and aging, these protective mechanisms are compromised, leading to tumorigenesis.
Genetic instability is one cause of disruption of these mechanisms. It is commonly present in both prokaryotes and eukaryotes.
For instance, in bacteria, it plays a significantly crucial role. In bacteria, genetic instability leads to the acquisition of new traits that provide them with antibiotic resistance and pathogenicity.
However, in multicellular organisms, including humans, it doesn’t have any positive role. For instance, genetic instability in humans is involved in carcinogenesis, several neurodegenerative disorders such as myotonic dystrophy, and ageing.
Douglas Hanahan and Robert A Weinberg described the eight hallmarks of cancer. They explained “genome instability” as one of the cancer hallmarks and explained its central role in tumorigenesis.
Genetic instability has a significant role in cancer. Genetic factors contribute to cancer, it is scientifically proven now! Since the 19th century, scientists have known that genetic mutations cause cancer. Genetic instability is one such type of alterations that highly contribute to tumorigenesis.
Genetic instability is also considered one of the key characteristics of aging. Interestingly, genetic instability and ageing have a bidirectional relationship. Aging is a natural process in which the body’s ability to function decreases, typically leading to increased disease vulnerability.
Meaning, as the body ages, it becomes susceptible to acquiring genetic instability. Vice versa is also true. As our genome becomes unstable, the accumulation of random mutations, DNA damage, telomere shortening, and epigenetic modifications declines cellular functionality, leading to aging.
Mutations and aging can lead to the activation of oncogenes and the inactivation of tumor suppressor genes, disrupting cellular homeostasis and leading to conditions such as cancer.
Scientists now know that various genetic or genomic instability has a significant role in various types of cancer. Genetic instability, cancer, and aging— these three factors are highly linked with one another.
Definition:
As discussed, when a genome becomes unstable by any random event, it acquires mutations and results in serious genetic problems.
Let’s define the term more scientifically.
“An increased tendency or higher frequencies of mutations within the genome during cell division is known as Genetic instability.” This leads to conditions such as cancer, aging, and some neurodegenerative disorders.
Types:
Genetic instability is categorized into three main types: Chromosomal instability, nucleotide instability, and microsatellite instability.
Chromosomal instability:
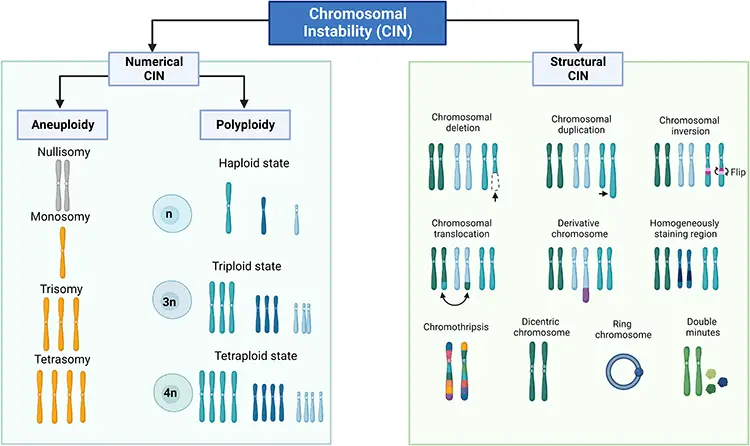
This is a type of genomic instability affecting the genome at the chromosome level. Chromosomal instabilities often result in conditions such as structural and numerical chromosomal alterations, including aneuploidies, polyploidies, deletions, duplications, insertions, inversions, and translocations.
Chromosomal instability is observed in two forms: Numerical CIN and Structural CIN.
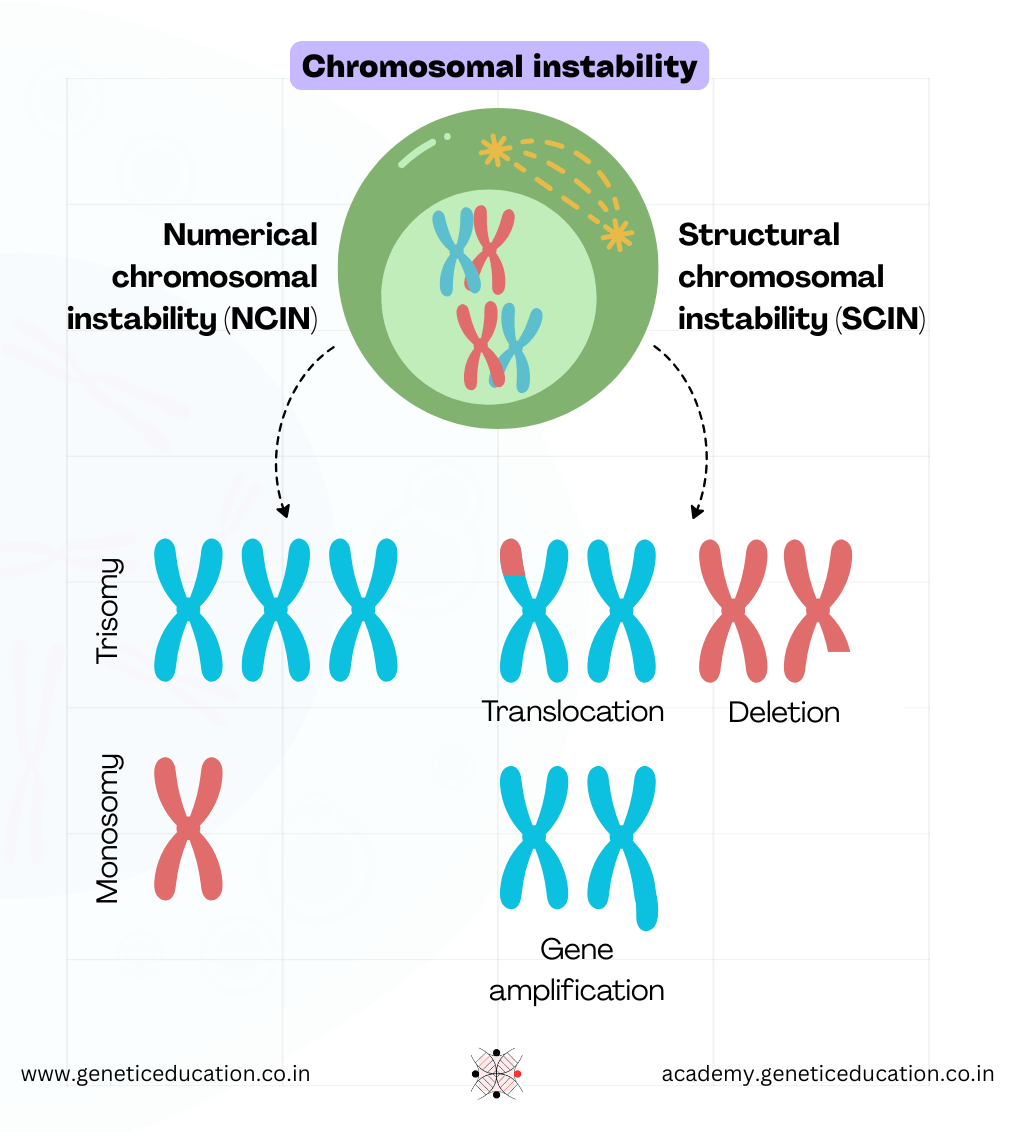
Errors in chromosome segregation result in numerical CIN. Numerical CIN does not change the nucleotide sequence within the chromosomes, it alters the number of chromosomes.
Meanwhile, structural CINs occur due to the gain or loss of chromosomal segments, resulting in alterations of nucleotide sequences.
Interestingly, it is observed that in the majority of tumor cells, structural CIN and Numerical CIN often coexist. Chromosomal instability is observed in 90% of solid tumors and also in blood cancers.
Nucleotide instability:
Nucleotide instability affects the genome at the nucleotide level. This type of instability results in nucleotide alterations, including deletions, insertions, and single-nucleotide polymorphisms.
Nucleotide instability is the least common but still contributes to several types of cancer.
Microsatellite instability:
Microsatellites are short repetitive DNA sequences scattered throughout the genome. Microsatellite instability occurs when these repetitive sequences start acquiring mutations.
Although such mutations are repaired by the mismatch repair system, when it fails, it results in microsatellite instability and leads to cancer. To know more about this topic, you can read our comprehensive article on this topic by clicking the link.
Telomere instability:
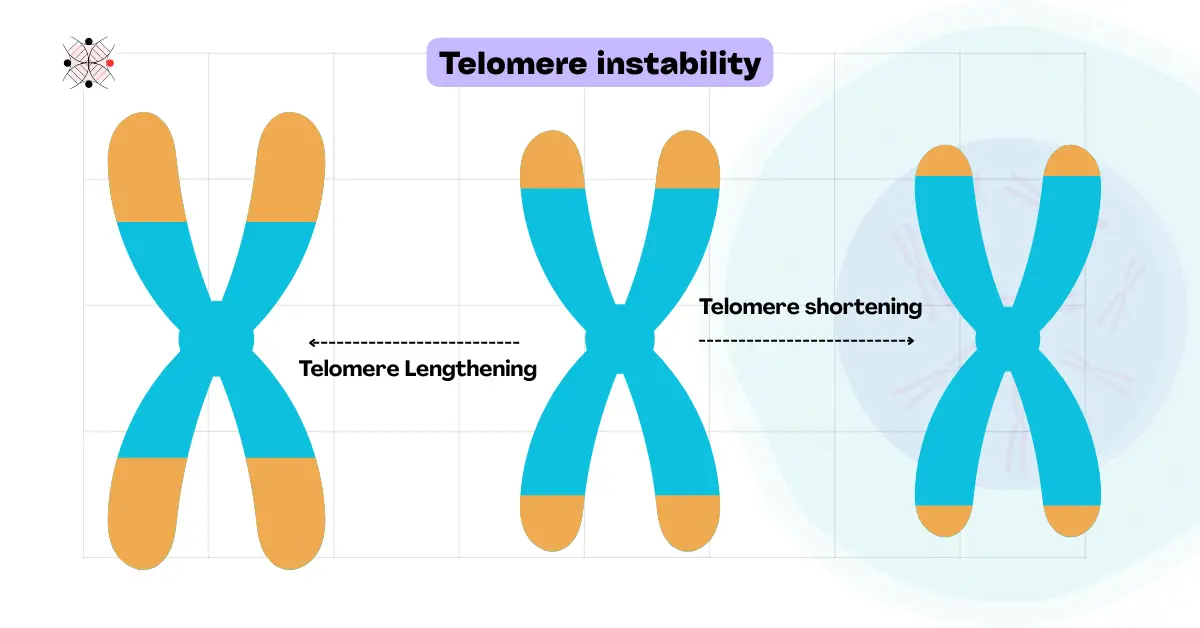
Telomeres are the protective chromosomal ends containing shorter repetitive DNA sequences. Telomere shortening or dysfunction usually causes telomere instability.
In consequence, it leads to chromosome or gene fusion, DNA damage, cell death, and cancer. Studies suggest a significant role in telomere instability in premature ageing as well.
Common factors contributing to telomere instability are aging, mutations in telomeric genes (TERC or TERT), oxidative stress, or abnormal telomeric replication.
Common examples are Werner syndrome (premature aging) and cancer.
Centromeric instability:
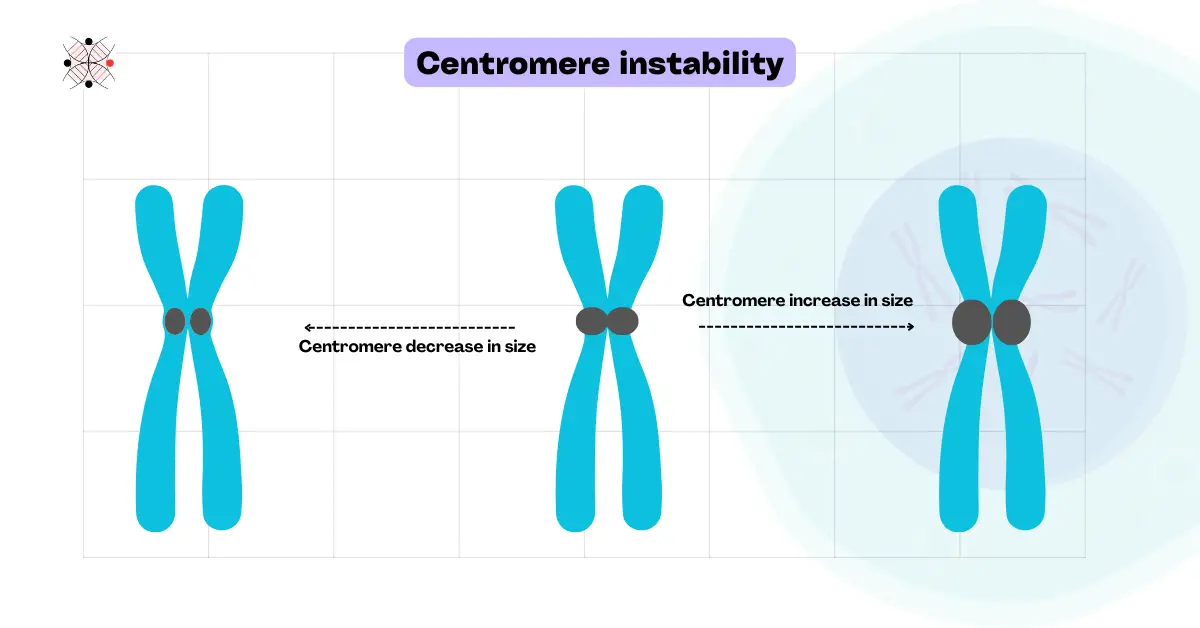
The centromere is the central chromosomal part. A dark and highly repetitive, gene-less region, where the spindle fiber attaches during the replication process and helps in the proper segregation of chromosomes.
Instability in the centromere makes it defective and leads to improper chromosomal segregation. Centromere instability thus results in one of the hallmark cancer alterations– aneuploidy (Abnormal chromosomal numbers).
With Radiation, chemical or carcinogen exposure being the most common causes, epigenetic changes in CENP-A or CENP-B, defective spindle assembly and several chromosomal abnormalities (like Robertsonian translocation) cause centromeric instability too.
It is also reported in various types of cancer.
Mechanisms of Genetic Instability:
Mechanisms of genetic instability involve complex interactions between genetic, epigenetic, and environmental factors. Let’s discuss some key mechanisms of genetic instabilities.
Defects during DNA Repair:
DNA repair pathways play an important role in maintaining genomic integrity. If the DNA repair pathways don’t work properly, it results in the accumulation of DNA damage, leading to genomic instability.
In normal functioning cells, DNA damages are repaired by several DNA damage repair pathways, such as the Direct reversal pathway, the Mismatch repair pathway, the Nucleotide excision repair pathway, the Base excision repair pathway, the Homologous recombination pathway, and the Non-homologous end-joining pathway.
If these repair mechanisms are impaired, it can result in genomic instability and give rise to conditions like cancer. In the table below, some of the examples are discussed:
| Type of Repair Pathway | Gene Affected | Type of Instability Resulted | Associated Cancers |
| Mismatch Repair (MMR) | MLH1, MSH2, MSH6, PMS2 | Microsatellite Instability (MSI) | Colorectal cancer, Endometrial cancer, Gastric cancer |
| Base Excision Repair (BER) | XRCC1, POLB | Point Mutations | Colorectal cancer, Breast cancer |
| Nucleotide Excision Repair (NER) | XPA, XPC, ERCC2 | Chromosomal Instability (CIN) | Skin cancer (e.g., xeroderma pigmentosum), Lung cancer |
| Homologous Recombination (HR) | BRCA1, BRCA2, ATM | Chromosomal Instability (CIN) | Hereditary breast and ovarian cancers, Prostate cancer, Pancreatic cancer |
| Non-Homologous End Joining (NHEJ) | XRCC5, XRCC6 | Chromosomal Instability (CIN) | Leukemia, Lymphoma |
| Fanconi Anemia Pathway | FANCA, FANCC, FANCD2 | Chromosomal Instability (CIN) | Leukemia, Squamous cell carcinoma, Breast cancer |
| Translesion Synthesis (TLS) | POLH | Point Mutations | Skin cancer (e.g., xeroderma pigmentosum variant) |
Errors in Chromosomal Segregation:
Meiosis and mitosis are two essential components of the human body by which cells can produce several copies of themselves.
Two types of cell division processes— meiosis and mitosis copy the DNA through replication.
Mitosis is the type of cell division by which cells produce two identical copies of the parent cell. This type of cell division is essential for growth and repair mechanisms.
While meiosis is the type of cell division by which cells produce four copies of a cell, these copies are non-identical daughter cells. This type of cell division is essential for egg and sperm cell formation.
Each of these cell division types involves the process of chromosome segregation. In mitosis, this is a single-step process in which chromosomes are duplicated, segregated, and form two copies of the cells.
While in meiosis, this process involves multiple steps such as synapsis, crossing over, and then segregation, eventually forming four non-identical daughter cells.
Improper chromosome segregation results in aneuploidy* (either too few or too many chromosomes) and is contributed to by genetic instability.
Several other mechanisms, such as telomere dysfunction, centrosome or gene amplification, and errors in kinetochore-microtubule attachment, lead to chromosomal instabilities as well.
In the table below, some of the examples are discussed:
| Mechanism of Chromosome Segregation | Gene Affected | Type of Instability Resulted | Associated Cancers |
| Centrosome Amplification | PLK4, AURKA, STIL | Chromosomal Instability (CIN) | Breast cancer, Lung cancer, Colorectal cancer |
| Kinetochore-Microtubule Attachment Errors | BUB1, BUBR1, MAD2 | Chromosomal Instability (CIN) | Colorectal cancer, Ovarian cancer, Pancreatic cancer |
| Telomere Dysfunction | TERT, TERC, POT1 | Chromosomal Instability (CIN) | Leukemia, Melanoma, Breast cancer |
| Spindle Assembly Checkpoint (SAC) Defects | BUB1, BUBR1, MAD2 | Chromosomal Instability (CIN) | Colorectal cancer, Ovarian cancer, Pancreatic cancer |
| Cohesin Complex Defects | SMC1A, SMC3, RAD21 | Chromosomal Instability (CIN) | Acute myeloid leukemia (AML), Colorectal cancer, Breast cancer |
Epigenetic Modifications:
Epigenetic modifications are essential for cells to regulate the process of gene expression and gene silencing. These are heritable changes that do not alter the sequence of DNA itself, but they can influence the process by which genes are expressed.
Among many, two key epigenetic mechanisms are DNA methylation and Histone modifications.
DNA methylation involves the addition of methyl groups to DNA at cytosine bases. Methylation of DNA is crucial for regulating gene expression as it can lead to gene silencing.
Histones are the proteins around which DNA is wrapped and form chromatin. Histone protein undergoes various modifications, including acetylation, methylation, phosphorylation, and ubiquitination.
Histone modification, such as acetylation, can loosen chromatin, which promotes gene expression. While modifications such as methylation can tighten chromatin, which promotes gene silencing.
Impaired histone modifications can lead to genomic instabilities by silencing tumor suppressor genes and activating oncogenes, which are responsible for several types of cancer. In the table below, some of the examples are discussed:
| Type of Epigenetic Modification | Gene Affected | Type of Instability Resulted | Associated Cancers |
| DNA Methylation | DNMT1, DNMT3A, DNMT3B | Chromosomal Instability (CIN) | Colorectal cancer, Leukemia, Breast cancer |
| Histone Acetylation | HDAC1, HDAC2, HDAC3 | Chromosomal Instability (CIN) | Prostate cancer, Lymphoma, Renal cell carcinoma |
| Histone Methylation | EZH2, KDM6A, KMT2D | Chromosomal Instability (CIN) | Prostate cancer, Lymphoma, Renal cell carcinoma |
| Chromatin Remodeling | ARID1A, SMARCA4, BRG1 | Chromosomal Instability (CIN) | Ovarian cancer, Lung cancer, Pancreatic cancer |
| Non-Coding RNA Regulation | DICER1, DROSHA, XIST | Chromosomal Instability (CIN) | Breast cancer, Ovarian cancer, Thyroid cancer |
DNA Damage by Oxidative Stress:
Oxidative stress is defined as “the imbalance between an organism’s ability to detoxify the reactive intermediates and the amount of Reactive Oxygen Species (ROS) it faces”.
Reactive Oxygen Species, such as free radicals, molecules capable of producing radicals, and several oxidizing agents, are capable of reacting with macromolecules, also capable of altering their structure directly or indirectly.
DNA damage due to oxidative stress can occur in two ways: either by endogenous factors or by exogenous agents.
Endogenous factors are spontaneous or enzymatic conversions and certain cellular processes that produce ROS, such as mitochondrial respiration and the inflammatory response.
Exogenous agents are air pollution, radiation, lifestyle, exposure to UV, and exposure to pesticides and metals.
Oxidative stress can cause various types of DNA damage, such as strand breaks, base modifications, and abasic sites. This type of DNA damage can result in genomic instabilities. In the table below, some of the examples are discussed:
| Type of DNA Damage | Exogenous Agents/Endogenous Factors | Gene Affected | Type of Instability Resulted | Associated Cancers |
| Base Modifications | Mitochondrial respiration, Inflammation | OGG1, MUTYH | Point Mutations | Colorectal cancer, Lung cancer |
| Single-Strand Breaks (SSBs) | Ionizing radiation, ROS | PARP1, XRCC1 | Chromosomal Instability | Breast cancer, Ovarian cancer |
| Double-Strand Breaks (DSBs) | Ionizing radiation, Chemotherapeutic drugs, ROS | BRCA1, BRCA2, ATM | Chromosomal Instability | Hereditary breast and ovarian cancers, Prostate cancer, Pancreatic cancer |
| DNA Crosslinks | Chemotherapeutic drugs (e.g., cisplatin), Aldehydes | FANCA, FANCC, FANCD2 | Chromosomal Instability | Leukemia, Squamous cell carcinoma, Breast cancer |
| Abasic Sites | Oxidative damage | APE1, POLB | Point Mutations | Colorectal cancer, Lung cancer |
Methods to Detect Genetic Instability:
Detection of genomic instability is crucial for cancer diagnosis. There are several methods for the detection of genomic instabilities. Some of the popular methods are discussed below:
Microsatellite Instability (MSI) Testing:
Microsatellite Instability Testing is a robust method for the detection of alterations in short, repetitive DNA sequences. These types of alterations most commonly occur due to errors in DNA Mismatch Repair Pathways. So this method is useful for the detection of faulty DNA mismatch repair mechanisms.
In this method lengths of microsatellite regions in tumor cells are compared with normal cells. By this comparison, the status of microsatellite instability is measured. High status or MSI-H denotes certain cancers and several genetic disorders.
This is a PCR-based method, in which the Bethesda panel is used, which includes five microsatellite markers. After microsatellite locus amplification, fragment analysis has been conducted by the Bethesda panel.
Karyotyping:
Karyotyping is a microscopic method used for the detection of chromosomal instabilities, which involves the visualization of chromosomes under a microscope. This method can easily detect structural and numerical abnormalities present in chromosomes.
This method involves the mammalian cell culture. Cultured cells are then harvested and followed by fixation. Then, slides are prepared and stained for microscopic visualization.
Karyotyping is a gold-standard method for the detection of aneuploidies. Hence, karyotyping can identify numerical and structural chromosomal instabilities.
However, it is unable to detect smaller chromosomal variations and balanced rearrangements. In addition, it can’t study solid tumors due to culture limitations.
Fluorescence In Situ Hybridization (FISH):
FISH (Fluorescence In Situ Hybridization) is an advanced technique to detect chromosomal instabilities. This method can detect alterations in the DNA sequence such as translocations, insertions, and deletions.
FISH is a probe-based method for the detection of various chromosomal abnormalities and instabilities. It has higher resolution than karyotyping and is routinely used in cancer diagnosis.
It can commonly investigate smaller chromosomal abnormalities, centromeric and telomeric instabilities. Nonetheless, it has several technical limitations and can’t investigate alterations smaller than 1 kb in size.
Next-Generation Sequencing (NGS):
NGS (Next-generation sequencing) is the most advanced method, which has revolutionized the detection of genomic instabilities with very high throughput and accuracy.
NGS can detect a wide range of alterations in the DNA sequences, such as point mutations, insertions, deletions, Copy Number Variations (CNVs), single-nucleotide polymorphisms (SNPs), and structural variations.
It is a massive parallel sequencing that enables scientists to investigate genetic instabilities across the entire genome.
Wrapping Up:
Genetic or genomic instability plays a crucial role in carcinogenesis. It can be detected using karyotyping; however, techniques like PCR, FISH, or sequencing provide more detailed analysis and confidence in results.
Genetic instability is a random event that occurs during any stage of an individual’s life and results in cancer. However, individuals having more exposure to radiation and carcinogens have higher chances of acquiring various types of genomic instability.
I hope you like this article. Do share and subscribe to Genetic Education.
References:
- American Cancer Society. (2022). Microsatellite instability (MSI) and mismatch repair (MMR) deficiency. https://www.cancer.org.
- Cortés-Ciriano, I., Lee, J. J. K., Xi, R., Jain, D., Jung, Y. L., Yang, L., Gordenin, D., Klimczak, L. J., Zhang, C. Z., Pellman, D. S., & Park, P. J. (2020). Comprehensive analysis of chromothripsis in 2,658 human cancers using whole-genome sequencing. Nature Genetics, 52(3), 331-341. https://doi.org/10.1038/s41588-019-0576-7.
- Hanahan, D., & Weinberg, R. A. (2011). Hallmarks of cancer: The next generation. Cell, 144(5), 646-674. https://doi.org/10.1016/j.cell.2011.02.013.
- Helleday, T., Eshtad, S., & Nik-Zainal, S. (2014). Mechanisms underlying mutational signatures in human cancers. Nature Reviews Genetics, 15(9), 585-598. https://doi.org/10.1038/nrg3729.
- Kandoth, C., McLellan, M. D., Vandin, F., Ye, K., Niu, B., Lu, C., Xie, M., Zhang, Q., McMichael, J. F., Wyczalkowski, M. A., Leiserson, M. D. M., Miller, C. A., Welch, J. S., Walter, M. J., Wendl, M. C., Ley, T. J., Wilson, R. K., Raphael, B. J., & Ding, L. (2013). Mutational landscape and significance across 12 major cancer types. Nature, 502(7471), 333-339. https://doi.org/10.1038/nature12634.
- National Comprehensive Cancer Network. (2023). NCCN guidelines for detection of genetic instability in solid tumors. https://www.nccn.org.
- Nowell, P. C. (1976). The clonal evolution of tumor cell populations. Science, 194(4260), 23-28. https://doi.org/10.1126/science.959840.
- Stratton, M. R., Campbell, P. J., & Futreal, P. A. (2009). The cancer genome. Nature, 458(7239), 719-724. https://doi.org/10.1038/nature07943.
- Telli, M. L., Timms, K. M., Reid, J., Hennessy, B., Mills, G. B., Jensen, K. C., Szallasi, Z., Barry, W. T., Winer, E. P., Tung, N. M., Isakoff, S. J., Ryan, P. D., Greene-Colozzi, A., Gutin, A., Sangale, Z., Iliev, D., Neff, C., Abkevich, V., Jones, J. T., … Ford, J. M. (2016). Homologous recombination deficiency (HRD) score predicts response to platinum-containing neoadjuvant chemotherapy in patients with triple-negative breast cancer. Clinical Cancer Research, 22(15), 3764-3773. https://doi.org/10.1158/1078-0432.CCR-15-2477.
- Zhou, B. B. S., & Elledge, S. J. (2000). The DNA damage response: Putting checkpoints in perspective. Nature, 408(6811), 433-439. https://doi.org/10.1038/35044005.
- Gonzalez-Hunt, C. P., Wadhwa, M., & Sanders, L. H. (2018). DNA damage by oxidative stress: Measurement strategies for two genomes. Current Opinion in Toxicology, 7, 87–94. https://doi.org/10.1016/j.cotox.2017.11.001.
- Castellanos, G., Valbuena, D.S., Pérez, E., Villegas, V.E. and Rondón-Lagos, M. (2023). Chromosomal Instability as Enabling Feature and Central Hallmark of Breast Cancer. Breast Cancer: Targets and Therapy, Volume 15, pp.189–211. doi:https://doi.org/10.2147/bctt.s383759.

The way you broke this down is incredibly helpful.
This article offers an insightful look into genetic instability and its link to cancer and aging. The detailed explanation of its types and detection methods, especially NGS, highlights the critical role of genetic mutations in tumorigenesis. Understanding these mechanisms could significantly impact future cancer therapies. What are your thoughts on how these findings might shape cancer treatment approaches?