“Taq DNA polymerase is renowned for its heat-stable properties. But do you know it’s extensively used in PCR experiments due to these other properties? Let’s check it out.”
Taq DNA polymerase is a polymerase enzyme that catalyzes DNA synthesis activities. Meaning, it copies the DNA strand. Taq is derived from the thermostable bacteria isolated from the Hot Springs at Yellowstone National Park.
The function of Taq DNA polymerase is to add nucleotides at a growing DNA strand using the primer-DNA junction as a template. It’s extensively used during Polymerase Chain Reaction— a technique to amplify the DNA.
Here are the 6 amazing properties of Taq DNA polymerase which support why it is used in PCR.
Stay tuned.
Disclaimer: Information provided here is collected from peer-reviewed resources and re-presented in an understandable language. All the sources are enlisted at the end of the article.
Key Topics:
6 Properties of Taq DNA polymerase:
Thermostability, 5 to 3’ polymerization, lack of 3’ to 5’ proofreading, stable activity, flexibility & costumizability and pH are 6 important properties of Taq DNA polymerase that give it recognition to use in the PCR.
Read more: A Guide to Choose DNA Polymerase for Your Experiment.
Let’s explain each, one after another.
Thermostability:
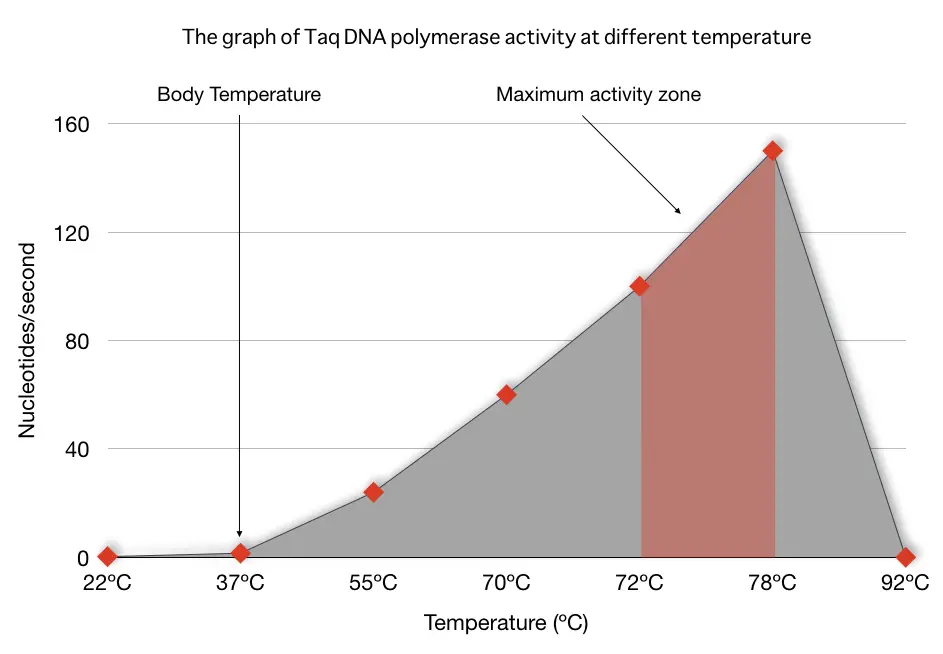
The most important and pivotal property of the Taq DNA polymerase is thermostability/heat-stability/heat-resistance. Common polymerases, in fact, all other enzymes work efficiently at body temperature or nearer (~37ºC).
PCR is a temperature-dependent enzymatic reaction having the highest and lowest temperatures of 90 to 98ºC and ~55ºC, respectively. Thus, our normal DNA polymerase is excluded from the race automatically to use here.
Taq DNA polymerase has the unique property of working efficiently at a very high temperature. It works skillfully between the temperature 70 to 78ºC, that’s the temperature of our PCR extension step.
In addition, during other PCR steps like denaturation (90 to 95ºC) and annealing (55 to 65ºC), it remains stable. Therefore, it’s the first choice for PCR amplification reactions. The Taq DNA pol. activity vs. temperature graph is explained here.
5’ to 3’ Polymerization:
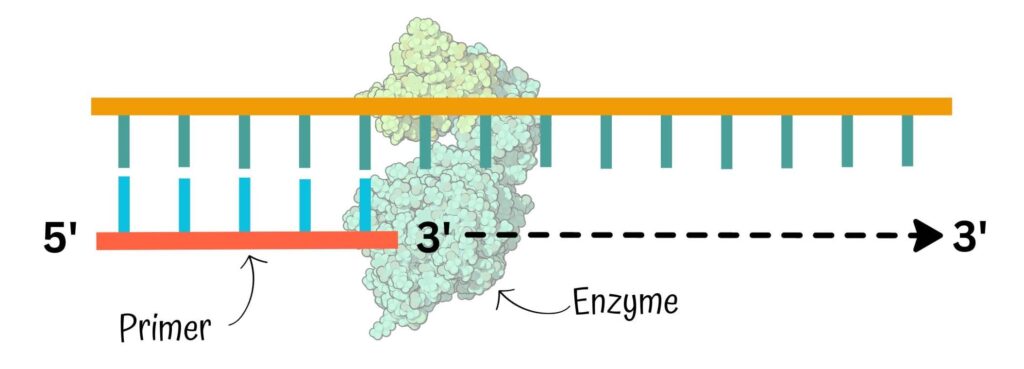
Taq DNA polymerase catalyzes the reaction only from a 5’ to 3’ direction. That’s exactly what we require. Primers provide a free 3’ OH end, which is the utmost requirement for Taq DNA polymerase to initiate synthesis.
Primers find their complementary location on the single-stranded DNA template, settle there and anneal. Now, the Taq DNA polymerase recognizes the primer-DNA junction and uses the free 3’ OH end as a starting point for synthesis.
It starts adding nucleotide to this 3’OH end. So its 5’ to 3’ polymerization activity has a crucial importance in the PCR. Check out the image below to better understand the concept.
Read more: Multifunctional DNA Polymerase.
Lack of 5’ to 3’ Proofreading activity:
Now, if you carefully read our previous article, you know that DNA polymerase has two types of activities— Polymerization and proofreading.
Put simply, the polymerization or synthesis domain governs the polymerization activity and adds nucleotide to the growing stand while the proofreading or exonuclease domain governs the proofreading activity and corrects any mis-base pairing.
We only want millions of DNA copies, that’s it! In PCR, we don’t want to correct any wrong base pairing. The presence of 5’ to 3’ proofreading activity can slow down the polymerization process and also decrease the enzyme’s efficiency.
Taq DNA polymerase is the best choice for PCR because it natively lacks 5’ to 3’ proofreading activity.
Stable activity:
Heat stable activity and lack of 5’ to 3’ proofreading activity are the two reasons, the Taq DNA polymerase can work stably during any step or stage of the PCR. For PCR amplification, it’s crucial that the enzyme works stably during denaturation, annealing and extension. Our Taq is capable enough to do that!
It can add up to 150 nucleotides/second at 72ºC, meaning, It can synthesize approximately 1.2Kb sequence in a minute. Here is one important PCR hack! If your sequence or target DNA amplicon is between 800 to 1000 bp, set the extension for 45 seconds only. That can save time and cost.
Flexibility and customizable:
Taq is a flexible enzyme and can be used with other enzymes as well. For example, it can be used along with the pfu DNA polymerase to increase the polymerization rate. The reason is, it actively can not interfere with other enzyme’s activity.
In addition, it can be customized as per the requirement. For example, the hot start DNA polymerase is an advanced version of the Taq DNA polymerase that can only work at higher temperatures.
Here, the Taq is linked with other enzymes or ligands to make it early-inactive. Taq lights up only when a higher temperature is applied (denaturation)– the ligand or enzyme is detached and Taq is added to the reaction.
Hot start PCR improves the accuracy of PCR by reducing potential non-specific amplifications.
pH of Taq DNA polymerase:
A PCR reaction buffer provides a stable 8.5 to 9 pH environment for the PCR reaction to occur. Taq works efficiently at pH ~9.0 which means no modification is required to use it in the reaction.
It mixes up with the ~8.5 to 9.0 pH environment and works efficiently during the amplification.
Read more: Comparison Between: DNA Polymerase vs RNA Polymerase.
Wrapping up:
Taq DNA polymerase is a crucial molecule in genetic experiments starting from PCR to DNA sequencing. However, other ingredients are equally important. It’s pivotal to know that inadequate Taq DNA polymerase can ruin the PCR reaction as well.
Thus, ideal storage conditions, accurate concentration & buffering conditions and a good reaction environment are required to make Taq work efficiently. If you want to learn more about PCR, you can read our previous articles or join our online course.
I hope you like this article.
Sources:
Ishino S, Ishino Y. DNA polymerases as useful reagents for biotechnology – the history of developmental research in the field. Front Microbiol. 2014 Aug 29;5:465. doi: 10.3389/fmicb.2014.00465. PMID: 25221550; PMCID: PMC4148896.

