Immuno PCR/IPCR/qIPCR is a “combination of the efficiency of RT-PCR and specificity of the ELISA method”.
As the name suggests the method is a combination of immunology and genetics. The method is based on the ELISA detection of protein and the real-time PCR analysis of DNA.
The immuno-PCR is also called IPCR or quantitative immuno-PCR/ qIPCR.
The ELISA is used for the protein detection and the RT-PCR is used for the detection of the DNA, then How the combination of both methods is possible?
Due to one of the major limitations of the ELISA method, the immuno-PCR technique evolved to overcome the problem.
The limitation is “low sensitivity in the detection of the lower number of analytes”.
In this article, we will understand How the RT PCR method supports overcoming this problem, what is the present scenario in the immuno-PCR and what is the future of immuno-PCR in diagnostics.
“With the help of the signal amplification capacity of the real-time PCR and the specificity of the ELISA, the immuno-PCR facilitates extremely powerful immunodetection method for quantitative detection of proteins.”
For understanding the immuno-PCR properly, we have to understand both techniques.
Let us understand the ELISA and RT-PCR broadly.
Key Topics:
What is ELISA?
ELISA is an enzyme-linked immunosorbent assay, EIA. ELISA is an immunological test used for the detection of antibodies present in a particular sample. The antibodies are the proteins released in the certain condition in which the antibody-specific antigens are present.
The sample is taken and the quantity of the antibodies present in the sample is detected using the ELISA, and ultimately the number of antigens present in the sample is determined.
The antigens are captured or sandwiched between the two primary and the secondary antibodies. By doing this, the concentration of a particular antigen can be detected.
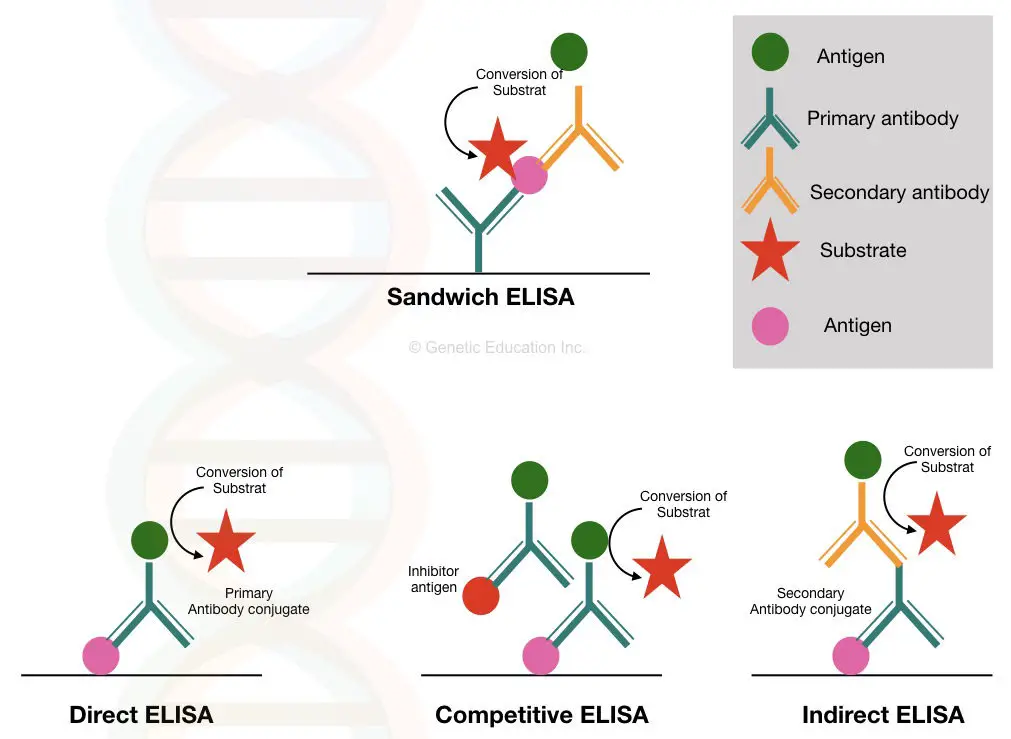
Indirect ELISA, direct ELISA competitive ELISA and sandwich ELISA are some of the modifications used for the detection of different types of antibodies.
Read more on PCR,
A Complete Guide to the Polymerase Chain Reaction
The image below is the graphical representation of the different types of ELISA methods.
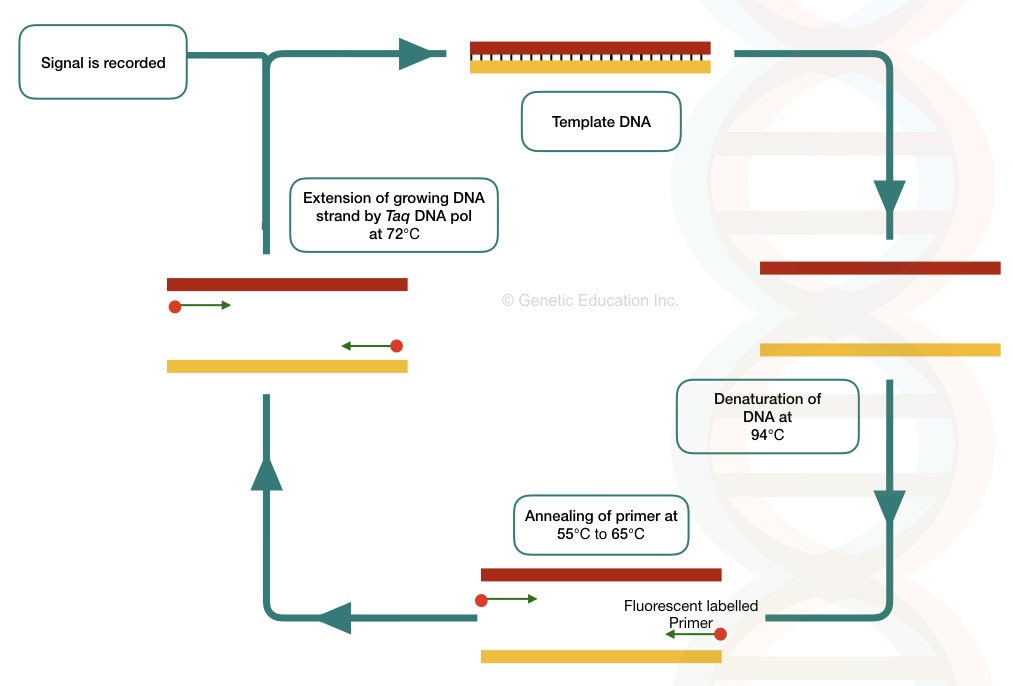
What is RT-PCR?
The labeled, single-stranded, short oligonucleotide primers amplify the DNA with the help of the Taq DNA polymerase and dNTPs which can be detected in the real-time data by the detector.
In the conventional PCR, the amplicons are detected or visualized on the gel but we can not determine how many amplicons are generated during the PCR. while in the real-time PCR we can calculate the exact number of the amplicons.
Never confuse Real-time PCR with reverse transcriptase PCR. The Real-time PCR is denoted as RT-PCR.
Read more on agarose gel electrophoresis,
Principle of the immuno-PCR/IPCR
The immuno-PCR method is developed in the year, 1992 by T Sano, L Smith and R Cantor.
The primary antibody immobilized on the surface of the plate binds with the conjugates that facilitate immunoreaction as well as the amplification reaction.
The “Conjugate” mentioned here, is the secondary antibody that is bound/conjugated with the oligonucleotide primer used in the PCR reaction.
The antibody-bound oligonucleotides amplify the DNA and are detected. The graphical representation is shown in the figure below,
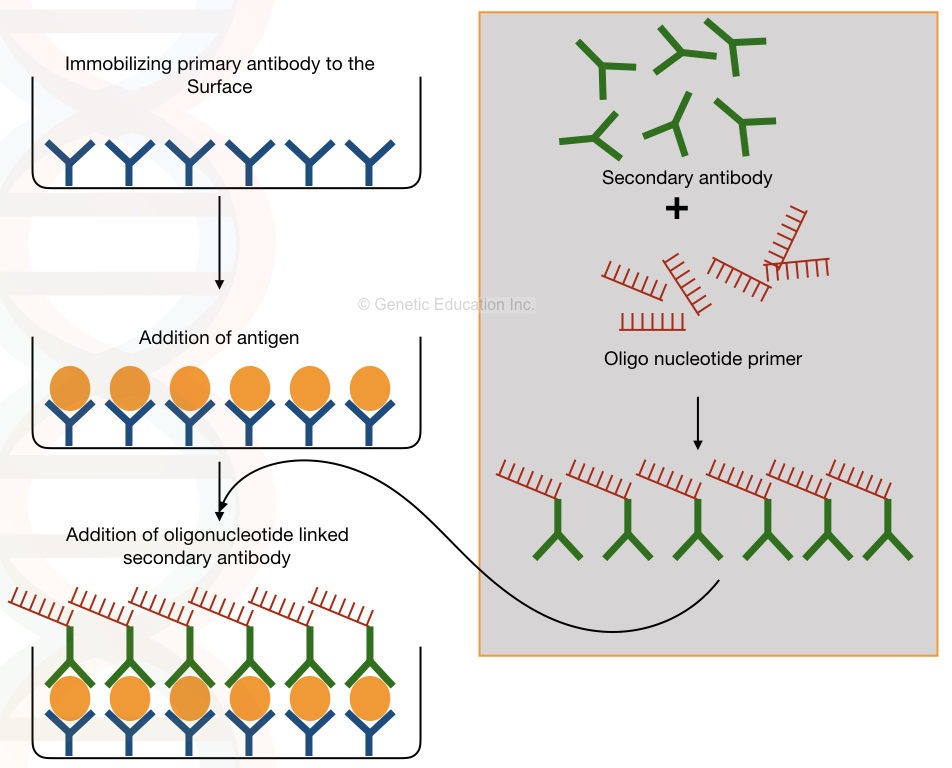
Process of immuno-PCR
The steps of the immuno-PCR/ IPCR are described in the figure below.
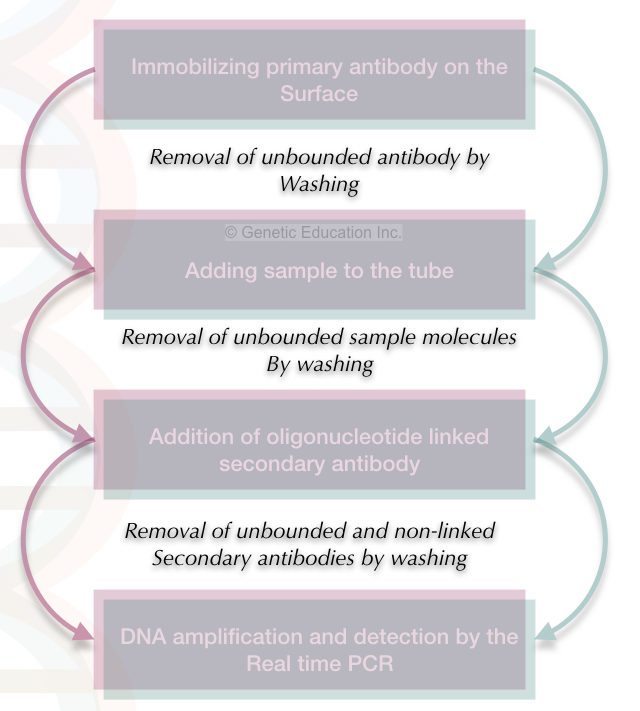
In the first step, we have to immobilize the primary antibody to the surface of the reaction tube. The step is the same as in the ELISA.
The non-bounded primary antibodies are washed off with the help of the washing buffer.
In the second step, the sample is added to the tube.
In this step, the sample molecules are bound to the primary antibody and make the conjugate. The unbounded sample molecules are washed off by the washing buffer.
After that, the oligonucleotide linked or the primer-linked secondary antibody is added to the tube.
In the second step, it immediately reacts with the secondary antibody-oligonucleotide-linked conjugate.
The excess conjugates are removed by washing.
Now our sample is ready for amplification.
Here the oligonucleotide primers are fluorescent-labeled. Hence it is detected during the PCR.
In each cycle, the primes bind with the template DNA and amplify it, the amplification is detected by the machine and recorded.
The amplicons are determined and so is the antigen which is conjugated with it.
Ultimately, the sample is quantitatively determined.
Read more on our immuno-genetics articles:
Benefits of Immuno PCR
The immuno-PCR facilitates the detection of an extremely low level of antigen which is not possible in the traditional ELISA. We can even detect a picogram of the concentration of particular analytes present in the sample.
The method is rapid and time-efficient. A lot of incubation steps are required in the ELISA, which are not needed in the immuno-PCR. Hence it is fast, accurate and rapid.
The reproducibility is very high and the detection range is wider than the ELISA method.
Further, one can detect multiple samples at once.
Limitations of Immuno-PCR
Conjugating oligonucleotides with the antibody required time and expertise.
The conjugation process is time-consuming, tedious and hard. Also, the cost of preparing a conjugate is very high.
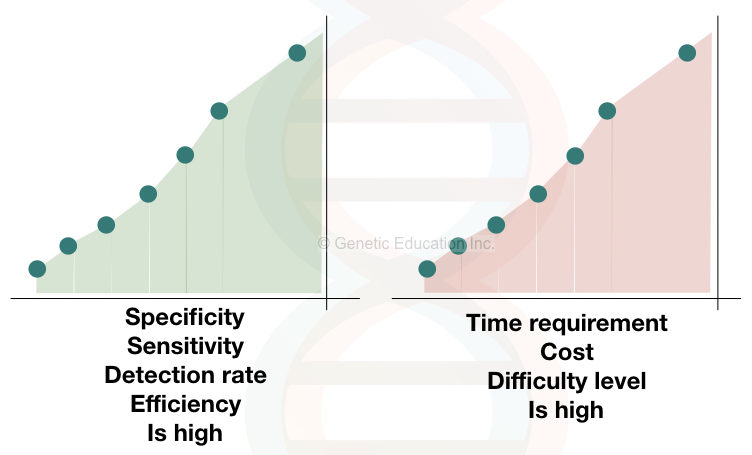
Albeit, the IPCR technique has power and potential, advancement and automation are not available right now. The specificity and sensitivity of the IPCR or qIPCR are 1000-fold higher than the ELISA method.
Furthermore, fewer research data are available on the IPCR. The success rate of conjugate preparation is very low and a large portion of the oligonucleotide primers are washed off during the washing.
This is the reason why it is time-consuming and costly. And because of that scientists are not interested in the automation of immuno-PCR. Here is the source of the original research paper of the scientist who discovered the method.
Wrapping up:
Although The IPCR or the immuno-PCR method has high specificity and sensitivity, still it is not used in routine diagnosis. Both the methods ELISA and RT PCR are gold-standard procedures in the disease diagnosis but the combination of both the methods is not a good option for the medical science.
Nonetheless, the immuno-PCR method is the best example of the “out-of-the-box thinking” and innovative spirit of the scientist.