“Chinese scientists had used CRISPR-like tools to edit the live embryo. By editing germ cells they had produced genetically altered babies and who are known as CRISPR-babies.”
The story of CRISPR-CAS9 has come up again after Emmanuelle C and Jennifer A. D had won the Nobel Prize for optimizing the efficiency of CRISPR-Cas9. Their findings of tracrRNA(Trans-activating crRNA) have changed the whole CRISPR scenario.
Gene Editing is not new to the world, was just a fairy tale a few decades ago, although. If we put conspiracy theories related to gene therapy and CRISPR aside, scientifically it’s a true gem that can be operated to cure disease, treat genetic abnormalities and repair faulty genes.
No one wants to produce superhumans or mutants that create uncertainty for mankind but ethical concern, for sure, is a priority for gene therapy experiments. Several problems need to be addressed before it will be commercially available much like the ethical concern first, and drawbacks, limitations, and impact on mankind obviously.
With many CRISPR-CAS9 tools approved and available by FDA, yet its applications and use for germline cells, and altering embryos is controversial. There are issues that should be elucidated first before it’s commercial use.
Briefly, gene therapy is of two types, somatic cell gene therapies and germline gene therapies.
When somatic cells are altered or their genetic composition is manipulated, which is restricted to a specific tissue, it is referred to as somatic cell gene therapy. Fair enough! can be employed for cancer-like treatments.
But when it is operated to alter the genome of germ cells, which will be going to make a whole new life, the change will be inherited to the new organism, big problem! Can’t be addressed. Scientists don’t advise germ-line therapies due to this reason and therefore it’s almost banned by every country.
In 2019, a controversy of CRISPR-babies arose when some Chinese scientists had successfully designed the CRISPR-induced twin babies in the lab. Jinakui and co-workers were accused of altering a human embryo, which is ethically and legally outlawed.
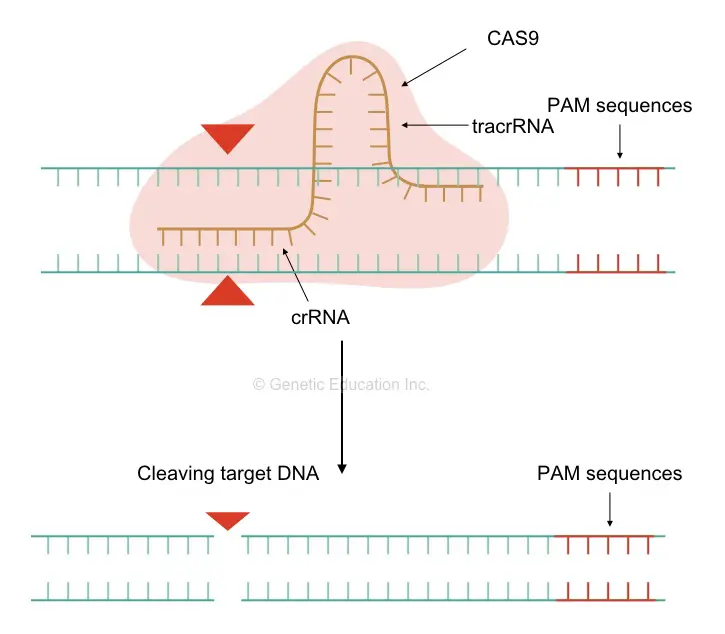
The CRISPR-CAS technique is a tool for gene editing, having derived from a bacterial defense system. A CRISPR- Clustered Regularly Interspaced Short Palindromic Repeats is a DNA sequence that helps Cas in recognition of the target location, which we wish to alter.
Here, the CAS9 or any other CASes are the protein, a kind of nuclease that cleaves DNA at a location where the CRISPR complementary crRNA binds. The Cas in the system recruits a special type of RNA known as the guided RNA, which is attached to the site, recognizable to Cas for cleaving.
Once the scissor- Cas, cleaved DNA at the location we wish to study, we can insert or remove a gene from that location. That is how the whole mechanism works!
CRISPR-babies and He Jiankui’s case:
He Jiankui was the first person who had used the CRISPR gene-editing system to edit the live human embryo through in vitro fertilization technique, working at the Southern University of Science and Technology, China. The controversy came to light when he had uploaded a series of youtube videos on his case success.
In 2017, a Chinese couple with an HIV-positive father and HIV-negative mother was offered an in vitro fertilization with Gene editing to assure resistance against HIV, under the guidance of He J.
Technically, using the CRISPR concept, they had actually edited the genome of the live embryo for altering the CCR5 gene which was supposed to give resistance to a baby against HIV.
When this “secret project” got completed, He J had announced a successful birth of twin babies- the ‘CRISPR-edited babies’ born in October 2018.
IVF in a nutshell:
The IVF- in vitro fertilization technique, a hope for infertile couples, is an artificial technique to fertilize embryos in the lab. IVF is totally legal and advisable for couples facing infertility and fertilization problems.
Here the female egg is taken to fertilize by a healthy sperm in a lab condition. Once fertilization is completed, it is allowed to grow for some time and immediately inserted into the host female. It is a good technique to overcome infertility to some extent. Now let’s go back to our CRISPR-baby story.
Why CCR5 gene?
He J and co-workers had used the scientific data showing the role of CCR5 gene-encoded protein, having a definite role in allowing HIV entry in a cell. The protein encoded by CCR5 makes it possible for HIV to enter inside the cell.
However, some 10% of the European population whose CCR5 gene is mutated, are resistant to HIV to some extent.
Their concept was based on inserting a frameshift mutation to block the protein produced by mutating the CCR5 gene using the CAS9 mediated CRISPR tool.
A frameshift mutation is a kind of genetic alteration- a point mutation, shift or alter the entire reading frame when a base(nitrogenous) changed. Simply put, the reading frame is the portion that helps in making the coding part for a gene.
They had prepared a mosaic type of cell population with altered and unaltered cells and closely monitored the progress to identify the off-target effects by preimplantation genetic technique.
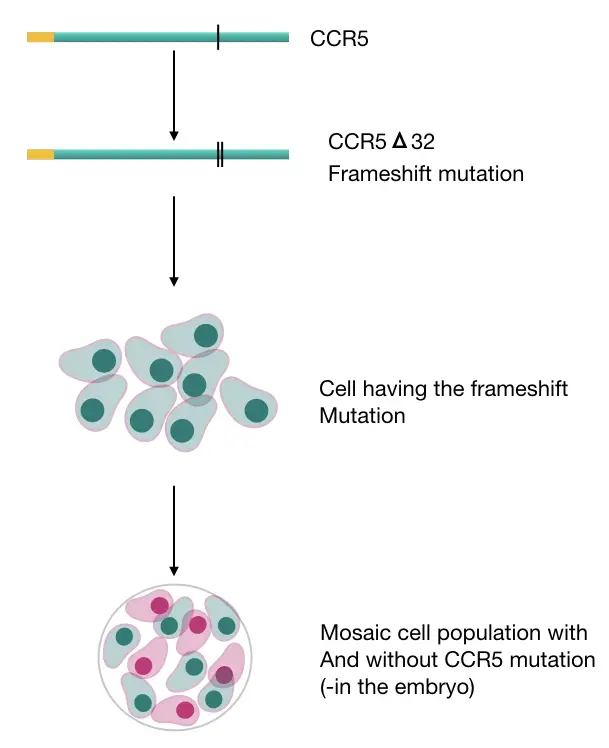
Following the pre-implantation genetic screening technique, they had sequenced mutant and non-mutant cell types.
The CRISPR technology is yet not fully ready to use for humans and to cure genetic disease, said Richard Lifton, President Rockefeller University, New York.
“CCR5 gene mutation may help those babies not only to fight against HIV but also from other infections like West Nile virus and makes them vulnerable against it.” said He J.
However, the court addressed that if babies might suffer from any related condition, it could be considered medically illegal and He J & co-workers could be found guilty in the case.
He J and the other two co-workers were sentenced to prison for 3 years for using the CRISPR-like germline gene therapies and not taking appropriate safety precautions & standard procedures for gene editing.
Total three CRISPR- edited babies were born in 2018 and 2019, a twin sister Lulu and Nana in 2018 and a third baby in 2019.
On 30 December 2019, Chinese authorities declared Hu J, Renli S and Jinzhou Q guilty for unethical use of the scientific data and fined 4,30,000 USD as well as 3 years prison sentence.
The work done by those scientists was criticized but was groundbreaking. The successful birth of a CRISPR-edited baby also had given hope to the scientific fraternity and proved that what beyond imagination, a human can do!
But the question is, what were the problems? What’s wrong with their research work? It is necessary for newbie scientists to understand what were the negative consequences of the present case study! Here are some of the points;
The role of CCR5 delta32 mutation…
The CCR5- C-C chemokine receptor is the protein where HIV binds and enters into a cell, the CCR5 gene is located on chromosome 3. The natural mutation of CCR5 delta32 in which 32 base pairs of the following gene got deleted can’t form a protein product and hence HIV can’t enter into the cell, through this receptor, conceptually.
The heterozygous mutation is less effective or its effect is negligible than the homozygous one. However, cases with resistance against the HIV infection by the homozygous CCR5 delta 32 mutations are rare.
The mutations induced through the CRISPR-CAS9 were basically not the original delta 32 one, one baby carried the 15 bp frameshift mutation while others carried a 4 base pair frameshift mutation.
In addition, the mutation they had induced was heterozygous, which was another scenario that actually couldn’t work.
One of the biggest flaws of the present technique is the off-target and the consequence of the present mutation in other regions of the genome.
Sometimes it happens that the CRISPR slips the recognition site and induces cleaving DNA other than the target location, consequently, other genes’ functions get altered. It might be 1, 2, 3 or more genes whose activity might be affected by the off-targeting effect.
Another negative aspect of the present technique was that it can adversely influence other regions of the genome Now, in the future or in future generations.
He j and his coworkers were unanswerable to this; they fail to explain the future effects of mutation introduced.
Nonetheless, studies on model organisms suggest that off-targeting and adverse effects of induced mutation are negligible. Scientists had reported no side effects, the harmful or lethal effects of more than 40 off-target mutations in the mice model.
Still, supportive evidence on human trials is no longer available.
Related article: what is gene knockout?
Unpublished research…
Every research should be published in a peer-review journal with prior ethical approval. He J and co-workers had submitted their manuscript entitled, “birth of twins after genome editing for HIV resistance” to Nature as well as Journals of American Medical Association.
However, both rejected their application for publishing their article due to serious ethical considerations and issues. Then he had presented their work, which was unpublished and had no scientific background.
Also, their host research institute Southern University of Science and Technology announced that the present research wasn’t done on their premises or even not under their regulations. It was performed outside the university.
That had created a major setback for He J group.
Ethical approval…
One should have to take prior ethical approval from a well-known organization before conducting research on human subjects. Ethical approval is compulsorily required in every country.
He J claimed that they had taken prior ethical permission from the Shenzhen HarMoniCare Women and Children’s hospital. Notably, the hospital denied having such types of records or research in their hospital.
In addition, though He J and colleagues were aware of the ethical consideration and issues, they still had performed such experiments without a medical practitioner in their team. They are biophysicists, not a medical person and therefore they can’t perform such experiments.
Tampering with ethics…
“Informed consent” is an important practice that must be followed by scientists even though they have the ethical approval or prior permission to experiment on human subjects.
Informed consent should be self-explanatory and must state the objectives and outcomes of the experiment to the subject or patient.
Evidence also suggests that He J and colleagues had taken informed consent unethically. The experiment was represented to the couple stating that the project was for AIDS vaccine development.
Gene-editing or CRISPR techniques or related things were not mentioned in the consent.
Against nature…
The negative point explains why He J referred to as China’s Dr. Frankenstein is altering the live…human… embryo, unethically.
Playing or experimenting on a live human fetus or embryo is actually considered against nature. Because consequences or long-term outcomes or drawbacks are not known to us.
What if it adversely influences the enreti mankind! God knows. We can’t predict, by altering the natural genome, what catastrophic effect it can produce.
CRISPR is amazing, indeed, if it can send someone to jail, how it is helpful, are there any benefits of it? The answer is yes, But it is not ready for now to use on human subjects.
We can edit genes
We can cure inherited genetic disorders
We can stop the spread of infectious diseases and thereby the spread of pathogens.
We can produce new useful traits and phenotypes
We can even repair the faulty genotype and phenotype as well
But all these are just the possible applications at least for now for humans. However, tremendous results are obtained in plants by the CRISPR-CAS9 gene-editing technique. You can read more here: Applications of CRISPR-CAS9 in Medical Science, Diagnostics, Research, Plant Biology and Agriculture.
Conclusion:
The case of CRISPR-babies and He J is a lesson for those crazy and extraordinary geniuses that scientific research must be performed in a proper scientific manner.
Prior permission, supportive evidence, and data, ethical concerns, personal concerns, informed consent must be taken priorly, showing the clear way of research and possible outcomes.
Sources:
The CRISPR-Baby scandal: what next for human gene-editing. David Syranoski Nature 566, 440-442 (2019).



Please crisper or genatics to cure all cancers treatments .??