Learn about PCR controls– Positive, Negative and internal controls, how to ensure accuracy & reliability, and why we use them in molecular genetic studies.
PCR (Polymerase Chain Reaction) is a technique to generate copies of DNA by amplification. Taq DNA polymerase, dNTPs, PCR primers, template DNA, PCR master mix and water are common PCR reaction ingredients. In molecular genetics, PCR is used to detect and amplify any DNA sequence.
However, PCR amplification is a highly error and contamination prone technique and leads to abnormal results and misinterpretations. And oftentimes, errors occur. So how can we overcome it? Or prevent misinterpretation of results?
PCR controls ensure the accuracy and reliability of any PCR reaction. Common PCR controls are positive and negative controls. But there are other controls used as well.
In this article, I will explain to you the concept of PCR controls including positive, negative and other common and uncommon controls used in the PCR reaction. The learning objectives of the present are to better understand and learn how to perform an effective and trustworthy PCR reaction.
Stay tuned,
Key Topics:
What is a PCR control?
PCR controls are used to determine the accuracy and reproducibility of the experiment. These eventually increase the trustworthiness of the experiment. Furthermore, it ensures the validity of the experimental procedure. Three common PCR controls are positive, negative and internal controls.
Besides, other unusual but useful controls are external control, template control and standard control. The inclusion of controls in the reaction tells us whether the reaction occurred correctly, if the results and reliable and trustable, and if the false positive or negative reaction occurred or not.
Controls in the PCR reaction work as a quality assurance that leads us to a conclusion about the success or failure of the experiment. Hence are an important part of any PCR reaction, especially when dealing with clinical samples.
Confusing? Let’s start with some basics.
PCR is a highly sensitive molecular genetic technique. Any small change in any parameter or factor leads to fatal errors in results. False positives and false negatives are two common types of errors in PCR. A list of parameters influencing any PCR reaction is enlisted here.
- The quality and quantity of ingredients.
- Reagent preparation scheme.
- Amplification conditions.
- PCR cycling conditions.
- The quality of Taq DNA polymerase.
- The time of PCR run.
- The composition of the PCR reaction buffer.
- The quality and quantity of template DNA.
- Lab conditions.
If any parameters dissolve, or conditions are compromised, it misleads the results. Such misleading results are,
- No amplification
- Poor amplification
- Primer-dimers.
Other than these, many other types of false results may appear in the PCR. For instance, amplification in a template-lack reaction is also a type of error. But how can we know? Whether the amplification is false, did we forget to add the template or not?
We can’t draw any conclusion, in this scenario, because we don’t have any reference, or quality control scheme to validate the results. Here, PCR control helps us to verify the results.
Note that the DNA ladder is also a type of reference using which we can validate the length of our amplicon, thereby, the success of amplification. Without a ladder or DNA marker, we can’t even say anything about the PCR results.
(It’s like looking for a travel destination without navigation.)
Coming back to our point– ‘Controls.’
Those— Controls are some pre-set parameters using which we will interpret our PCR results. PCR controls are used to
- Validate the performance of the test.
- Validate the results of the test.
- Evaluate reasons for false or negative results.
- Troubleshooting the problems.
- Give strength and scope to optimize other reactions.
So, conclusively, they give us the strength to verify the present results and scope to optimize future reactions by helping in troubleshooting. Now take a look at various types of PCR controls.
Types of PCR Controls:
Common types of PCR controls are positive control, negative control, internal PCR control, external PCR control, reaction control, PCR standard, etc. Let’s understand each, one after another.
Negative control:
This is often known as ‘negative amplification control (NAC)’ or no template control. It’s one of the simplest and most common PCR controls. It usually lacks the template DNA, meaning, while preparing the reaction, we do not add the template DNA.
A negative control is a reaction that does not contain our target or any template DNA. And used for the detection of contamination and false positive results. So in a gel, we can’t observe any bands in the negative control lane.
(No DNA, no amplification).

Instead of template DNA, nuclease-free water is added to the reaction which also validates whether the water is of high quality or not. Negative control tells us,
- If reagents are contaminated or not
- If any foreign DNA, viral DNA or naked DNA is accidentally incorporated in the reaction or not.
- If the nuclease-free water is of high quality or not.
- How to set up Negative control?
Add Taq DNA polymerase, forward and reverse primers, PCR buffer, and nuclease-free water. Place and run the reaction using the standard reaction protocol. We can also add PCR enhancers if they are used in the main reaction to validate the purity of enhancers as well.
- What is not included in the Negative control?
The negative control tube lacks “template DNA”.
As aforementioned, negative control has significant importance not only in conventional PCR but also in quantitative and reverse transcriptase PCR as well. In addition, keep one technical note in consideration. Primer dimers may appear in the negative control lane in the gel.
Because it lacks a template, not primers!
Occasionally, spurious primer amplification may be seen, which is not our target amplification. If it shows amplification (DNA band in a gel) of any size, any reagent may be contaminated. In that case, it’s advisable to use a fresh batch of reagents.
Positive Control:
Positive control shows reaction amplification. It is a reaction containing the template or target DNA sequence. Adding positive control in the reaction determines the sensitivity of the experiment– which means, everything is fine with the reaction or not. A positive control reaction is prepared with the same reagents and primers used for the experimental sample.
Standard positive control:
Standard positive control (SPC) is also a type of positive reaction containing artificially synthesized nucleotide sequences or available commercially with the kit. The reaction conditions and sample amplicon size of standard control are similar to our experimental sample.
This control is very crucial when in the case of multiple amplification bands. If our experiment sample shows multiple amplification bands, the presence of standard positive control helps validate the presence or absence of the correct amplicon in the reaction.
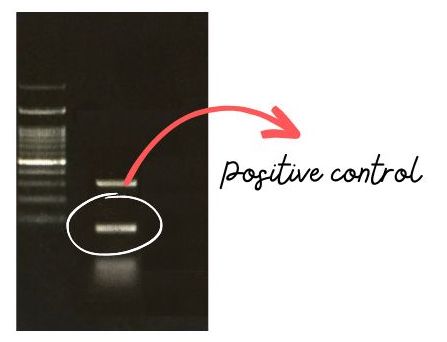
Conversely, if amplification would not occur in the standard control, make sure that there is something wrong with the reaction preparation step or PCR conditions. Usually, ready-to-use detection kits for sickle cell anemia, pathogen detection kits or any other amplification kits contain standard control.
- How to set up positive control?
A similar reaction to our experimental sample is prepared including Taq DNA polymerase, dNTPs, Primers, nuclease-water and the standard provided in the kit.
- What does it show?
It gives us an idea regarding the efficiency of the ingredients and reaction.
Note: Previous amplicons of the ‘same’ reaction can also be used as a standard positive control.
Internal control:
Internal control also known as internal positive control or internal amplification control (IAC) is a reaction-independent control. The internal control is designed in a way that co-amplifies the genomic region other than our target, using the same annealing temperature, reaction conditions and ingredients (except primers).
So technically, when we add internal control, we will get two bands in gel lanes, one for our experimental sample amplicon and another for internal control. Why do we do it?
It simply tells us if everything is going well with the experimental sample reaction or not. For example, let’s say we have prepared 10 sample tubes but don’t get amplification in the 9th tube. We don’t have any internal control added, can we say anything?
No.
We don’t know if we forget to add something to the reaction or not. Now, imagine that we have set up internal control. The presence of both internal control and experimental sample amplicons validate the results while the absence of either band shows that the reaction isn’t prepared well.
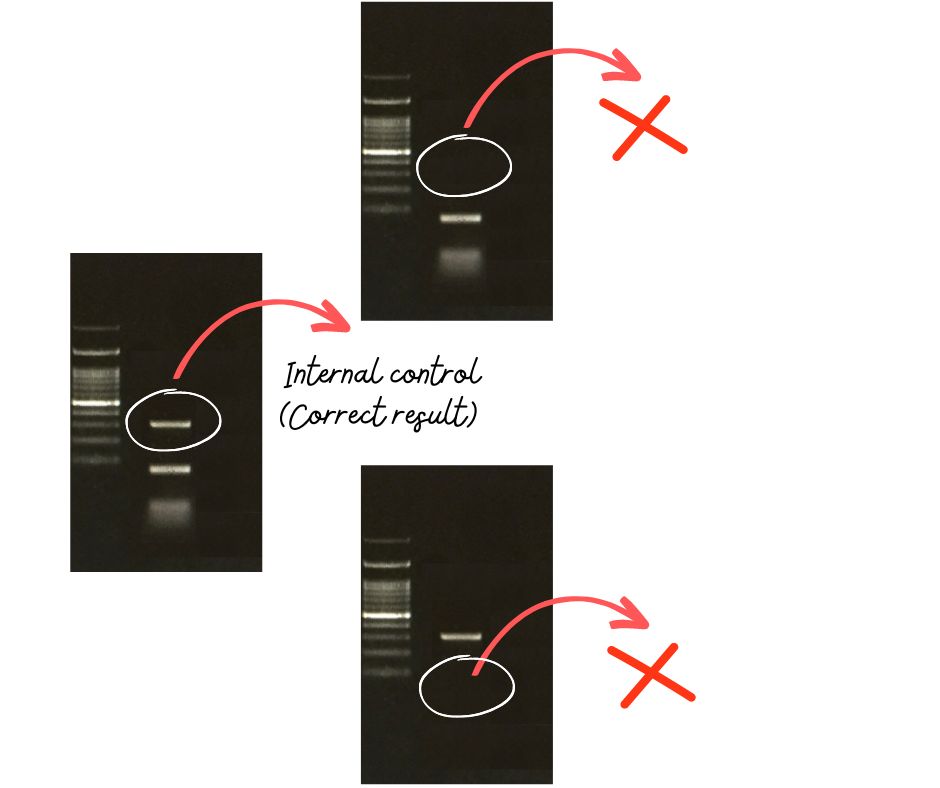
Such controls have a very important role in ARMS-PCR and the detection of single nucleotide polymorphism. However, keep in mind that multiplexing reactions may occasionally mislead the results.
Importantly these types of positive controls provide information related to reaction-specific or template-specific PCR inhibitors.
See the image below,
- How to set up internal control?
Add another set of off-target primers along with the standard reaction mixture– dNTPs, water and Taq DNA polymerase.
- What does it show?
It gives us an idea if our polymerase is working efficiently or not and if all the ingredients are utilized correctly or not.
Amplicon control:
Standard amplicon control is nothing but the amplicons of the previous reaction which doesn’t include in the PCR reaction but are directly added to the gel. It can’t tell us about PCR reaction or amplification conditions. However, it shows if there is something wrong with the gel preparation or not!
Notwithstanding, it’s very important to include it because sometimes it is just a simple gel preparation error that leads to tons of repeat experiments, cost and frustration.
Quantitative standard control:
RT-PCR and relevant assays usually need quality control or standard predetermined controls that form a base for the standard curve for quantification. Commonly, the quantity of these controls is known and provided by the manufacturer.
To understand the concept precisely let us take two examples one after another for PCR and qPCR.
Suppose we have to amplify one 300 bp product in our PCR. It’s an ARMS PCR assay meaning it utilizes 4 different primer sets to amplify different alleles. If you wish to learn more about ARMS PCR, click the link.
We have prepared a typical PCR reaction consisting of dNTP mix, Primer mix, Taq DNA polymerase, PCR reaction buffer, nuclease-free water and template, and put reactions at standard PCR cycling conditions.
Just look at the image below, it shows the results of our Hypothetical situation.
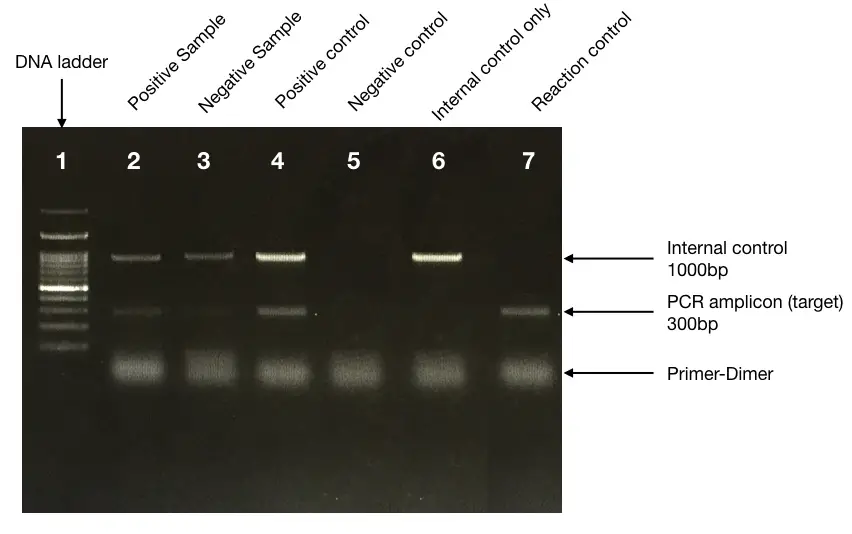
The first lane is our DNA ladder, the second lane consists of the PCR product which is the 300bp fragment- a normal sample. The third lane shows the results of the allele which is not amplified.
The 4th lane is our positive control. The fifth lane is the negative control showing nothing. The 6th lane is internal control without the template amplification.
The small band-like diffusion products are some primer dimers. Every ~1000 bp fragment in every lane is our internal control. So this is the basic setup for any PCR reaction. All these tubes and controls must be added to any PCR reaction.
Let us take an example of a typical SARS-CoV 2 RT-PCR kit which comprises three types of controls. Positive, negative and internal control.
As we said, negative control shows no amplification. It assures no contamination at any level of reaction preparation.
The positive control consists of the nucleotide sequence similar to our target SARS-CoV2 and shows amplification during the reaction. It also indicates that everything is working fine in the reaction.
Internal controls are of two types, the kit manufacturer provides one as per their SOP. The endogenous internal controls which are typically housekeeping genes amplify along with the template showing whether the starting template has a sufficient quantity or not.
The exogenous control is usually avoided to reduce the complexity of the assay. RT-PCR gives us results as the standard curve needs negative control, and positive quality control to determine results.
Any background signal during the reaction shows the presence of a contaminant. Besides providing quality assurance, positive control provides a reference value to estimate the quantity of the template too.
| Control | Ingredients | Template DNA | information |
| Negative control | Taq DNA polymerase, dNTPs, primer set, reaction buffer, nuclease-free water. | Absent | Validates reagents. Indicate contamination. |
| Positive control | Taq DNA polymerase, dNTPs, primer set, reaction buffer, nuclease-free water and template | Present | Validates Taq DNA polymerase activity. Provides reference value for the assay. |
| Internal control | Taq DNA polymerase, dNTPs, primer set, reaction buffer, nuclease-free water, template and internal control primers | Present | Check sufficiency of template validates the activity of Taq. |
Importance of PCR controls:
Now let’s answer the question, why do we use them in the reaction?
Quality control:
PCR controls are important quality control parameters in any molecular genetic experiment. It can ensure that the reaction is accurate, reproducible, and reliable. It also helps to identify potential errors and problems thereby helping in troubleshooting too.
Validation of reaction:
The inclusion of PCR controls also ensures that all the PCR ingredients are added to the reaction or not and shows the efficiency of each ingredient.
Validation of reaction conditions:
Annealing, temperature, PCR cycles, the concentration of primers, primer design and reaction time are crucial PCR reaction condition parameters that directly impact the reaction. The inclusion of PCR controls validates if the reaction conditions are adequate or not. And also ensures reaction efficiency and reproducibility.
Detection of contamination:
Any PCR reaction is highly prone to contamination by intrinsic and extrinsic factors. The use of negative or no template control helps determine any contamination present in the reaction. Hence it makes sure the accuracy and reliability of results.
Wrapping up:
Conclusively, every control I have discussed in this article should be included in a typical PCR reaction. However, the selection of control for PCR depends on the experiment being performed and the requirements of the experiment.
Put simply, PCR controls improve the sensitivity and accuracy of the results and also improve reproducibility. Do optimize your experiment before using it directly in the experiment sample. I hope this information is enough for the present topic.
Let me know if I miss anything. Do share the article and bookmark our page.



