“The science behind the probability of getting certain kinds of disease or finding the likelihood of getting, it is said as genetic predisposition.”
For the past couple of weeks, I was wondering to write a small piece of content on an interesting topic, “genetic predisposition”, the future of genetic studies, scientists believe.
The theory of genetic predisposition shows, “how often we acquire certain disorders”, mostly are chronic and polygenic disorders.
We have taken time to shape the content because the topic is boring, certainly and we can say a bit complex to understand for some group of students. We want to prepare content that is easy to understand and at the same time covers all the aspects. Now is the time, I can explain things in layman, hope you will like. Let’s start with some basic things.
Now what exactly the genetic predisposition is! In one line it’s a “likelihood of getting a disease” how often someone gets a disease or gets infected.
Until 1990, the field of genetic studies was restricted only to inherited diseases. Only those disorders which are inherited, having a definite genetic basis and whose phenotypes are well studied, were only classified into genetic disease and studied in genetic science.
Soon after around in 1990, the mechanism of DNA methylation was explained and a new era of genetics was evolved, “Epigenetics”, studies chances at gene expression level not at the molecular level. Now scientists would know that the genetic basis is much more explored than they thought.
After the discovery of genome-wide studies, genome sequencing and whole-genome microarray they have started correlating every minute life change, every single health condition with genetics. And the results were indeed great, every single health condition, disease or disorder has a definite genetic basis either at chromosome level or at the DNA level.
And after 2000, the concept of genetic predisposition came to light. Scientists were able to understand how chronic diseases occur.
Everything what we eat, where we live, what we are doing, our mental status, physical health, working place and our surroundings affect our DNA in many ways.
Some may change the expression of genes other may change the sequence. Changes or alterations are commonly known as “mutation”; which is either heritable or non-heritable, depending on which cells it occurs.
Key Topics:
What is genetic predisposition?
Genetic– the study of DNA or genome; Predisposition– tendency to get susceptible to some condition.
Definition:
“The science behind the probability of getting certain kinds of disease or finding the likelihood of getting, it is said as genetic predisposition.”
Genetic predisposition is a complex condition involving not only genetic basis (changes at DNA or chromosome level) but also intrinsic and extrinsic environmental factors too.
Disease, condition or disorders covering are complex, polygenic, non-inherited (mostly) and chronic. Meaning, many genes and alterations are there. By interacting with the environmental factors, it causes disease.
Trust me I don’t understand from where to start. I am typing things as it coming to my mind. Let us take an example before going ahead.
Suppose Your father is diabetic, suffering from type II diabetes- a common health condition worldwide. You are four offsprings; three are boys and a girl. Genetic predisposition shows your chances of getting diabetes type II, meaning, what is the probability that you will get diabetes from your father.
What scientists do! They took a sample from your father and study the whole genome, all alterations & SNPs and finds common ones. To correlate things, they take samples from all four siblings, again perform a DNA test, study the whole genome, all alterations and SNPs.
Now, they collect results and compare all four sibling’s DNA results with their father and finds who’s DNA profile is more likely similar to their father.
Supposed if your DNA profile is more matching with your father, your chances of getting diabetes from your father are more than the other three. However, it is also important to know that it doesn’t mean that you will get diabetes.
The example shows two important things related to genetic predisposition,
- First, the likelihood of getting the disease.
- Second, it isn’t a surety to get the disease.
List of disease:
Now first, let’s know which diseases are covered in the predisposition study. Here is the list,
- Type 2 diabetes
- Heart disease
- Cancer
- Obesity
- Autism spectrum disorder
- Stroke
- Asthma
- Bipolar disorders
- Autoimmune disease
- Lupus
- Celiac disease
- Schizophrenia
- Complex mental illness or mental retardation
- Multiple sclerosis chronic fatigue syndrome
- Rheumatoid arthritis
- Myopia
The influence of stressors and pathogen susceptibility is also genetically predisposed. -Sprangers M
Genetic predisposition vs inherited disease:
Genetically predisposed diseases are usually non-inherited, can’t pass down to offspring while inherited diseases pass down to their offspring in either autosomal dominant, autosomal recessive, X-linked dominant and X-linked recessive patterns.
Inherited diseases are single-gene disorders, usually, or a couple of genes or mutations are involved, while the other one is polygenic, many different variations exist, associated with the disease.
Reported genetic polymorphisms are said to be “associated with the disease” in the case of genetic predisposition, meaning it can’t directly cause the disease.
While in the case of hereditary diseases, the mutation or alteration directly causes the disease.
For instance, one with the mutation in the beta-globin gene may surely become either a thalassemia carrier or a thalassemia major.
Inherited diseases follow the mendelian pattern of inheritance whilst predisposition diseases follow the non-Mendelian pattern of inheritance.
| Genetically predisposed disease | Inherited disease |
| Type 2 diabetes
Heart disease Cancer Obesity Autism spectrum disorder Stroke Asthma Bipolar disorders Autoimmune disease Lupus Celiac disease Schizophrenia Complex mental illness or mental retardation Multiple sclerosis chronic fatigue syndrome Rheumatoid arthritis Myopia |
Sickle cell anemia
Thalassemia Huntington’s disease Cystic fibrosis Fabry disease Hereditary breast cancer Hemophilia Fragile X syndrome MTHFR gene mutations |
Mechanism of genetic predisposition:
The mechanism of predisposition works for non-mendelian/ non-inherited disease, you can explore the list above. Genetic predisposition studies are carried out using common genetic variations that are associated with the disease and present in the population. The study shows how someone is susceptible to certain diseases when came across some adverse environments.
For example, if your genetic test says that you have a 34% chance of getting some kind of cancer, you are living in the high UV radiations, the probability will surely increase.
Even, when you start consuming tobacco and related product or smoke, your chances of getting susceptible to cancer even increase more. That how the whole mechanism works.
It shows that not only the genetic factors but also other adverse conditions, environment, lifestyle and eating habits influence predisposition and occurrence of disease.
But on the safer side, if you know your chances of getting cancer is 34%, you avoid smoking & tobacco consumption and also stay away from UV & radiation areas, your chances of getting cancer will decrease.
Again, It isn’t guaranteed!!! The mechanism of genetic predisposition works on assumption and probability.
Genetic predisposition test:
The genetic predisposition test and process are different things, test performed individually while the process is followed to obtain and evaluate results.
The process:
DNA testing for genetic predisposition studies is conducted at the genomic level, meaning, whole genome, exons, common variants, common genes are studied, instead of a single gene or single variant.
Researchers do testing by collecting samples from the population, aiming to collect some common variant, data and information associated with the disease. Let say, our aim is to establish the genetic basis of breast cancer predisposition. To do so, we need to collect samples from breast cancer patients and their family members.
First, samples from breast cancer patients are processed and common genes, variants and mutations are identified.
In the next step, the frequency of every gene variant is measured, meaning, in how many patients a particular variant is present. Genes, variants and CNVs which are more common, are targeted to predisposition study. More accurate data are obtained as the sample size increases.
Based on the information collected, the frequency of predisposition is measured in other family members too. This is the whole process, following which researchers establish the genetic basis of predisposition.
Common methods to test are- whole-genome sequencing, whole-exome sequencing, SNP microarray, whole chromosome microarray, etc.
During the testing, scientists test and compare 1000 or more genes and variants from chronic disease patients with normal samples to establish the genetic basis.
Steps:
The process of testing initiates by collecting biological samples such as saliva, blood or other body fluid from patients and their family members.
Expert extracts DNA from samples, purify and process it for testing.
Based on the requirement of the assay and type of disease to be studied, the testing method is selected from the above-listed tests.
DNA sequencing is performed using the automated or semi-automated whole-genome sequencing technique.
Results are sent to the bioinformatics lab to know common genes, variants, SNPs and other mutations.
The probability of predisposition is decided based on the results.
We have learned only possibilities from the genome, how does 1 or 3% increased risk for something, let say cancer, translate into the clinic? That information is useless. -J Craig Venture.
Examples of genetic predisposition:
In this section, we will go through some of the studies conduction in this field.
Cancer:
Genetic predisposition of cancer though is complex but studied most commonly. Varied mechanisms, genes and variants are involved in causing a certain type of cancer.
More than ~290 different genes are constantly causing cancer at somatic and germ tissues, some are well studied and some aren’t! For example, the role and gene variants of the P53 gene are well-established in many cancer.
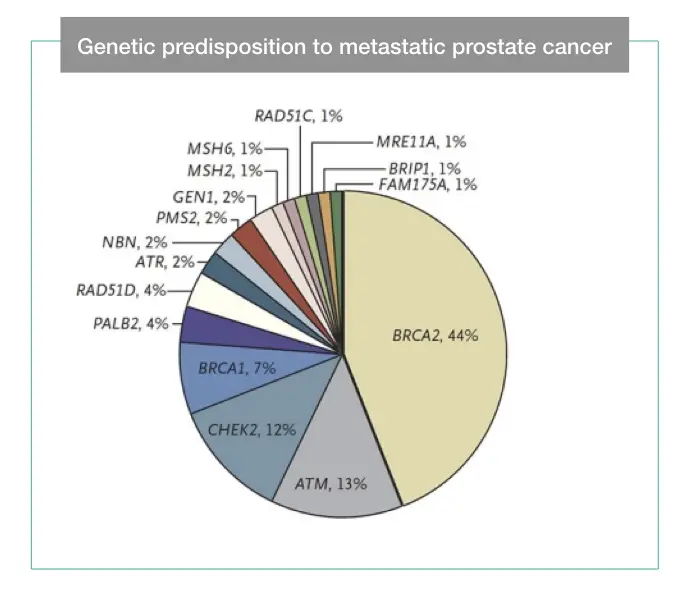
Some common genes are BRCA1 & 2, ATM, CHEK2, CDH1, STK11, TP53, BAP1, CDK4, MLH, PTEN, etc. Genes in cancer and further classified as tumor suppressor genes and oncogenes depending upon their activity.
Hamdi Y & Moujemaa M et al conducted a study on the Tunisian family showing the role of various genes in breast cancer using the technique of whole-exome sequencing.
Their finding suggests that other gene variants such as MMS19, DNAH3, KATB6 and POLK do have a definite role in breast cancer, particularly in this family.
Notedly, studies on monozygotic twins suggest that dissimilarities occur even in cases having similar genetic content.
That shows a clear role of environmental and epigenetic factors in the development of chronic disease. Meaning, the tendency of getting chronic cancer varies even in monozygotic twins. So cancer can’t be solely studied using genetic predisposition.
Adding to this explanation, some viruses and pathogens cause a serious type of cancer, damages genes and disrupt pathways. For example, let say pathogenic invasion produces A, B, C and D mutation linked to certain cancer.
If another person hasn’t infected with the same pathogen and still has the same mutations A, B and C, they can’t get cancer, ideally. However, predisposition studies show higher chances of getting cancer.
Note: Download this amazing Slideshare by Mahran Alnahmi: Genetic Predisposition to cancer Slideshare.
Schizophrenia and bipolar disorders:
Researchers using genetic susceptibility studies to explore conditions like schizophrenia, bipolar disorders and other psychological conditions. A study conducted in 2009, shows the involvement of more than 30,000 different gene variants in schizophrenia and bipolar disorders.
However, technically it is impossible to design a treatment or prevention program based on the data. Also, It is a time-consuming and costly process to screen and study all 30,000 gene variants.
Schizophrenia and related disorders are complex in nature, many genes and pathways are involved and therefore it is difficult to establish the genetic basis for those disorders.
Autoimmune disease:
Autoimmune disease occurs when our own immune system attacks our body. Again, it is a complex condition in which many genes, gene variants and environmental factors have a role.
Our immune system protects us from viral and bacterial infection using many different pathways and systems, however, because of some mutations, the whole mechanism gets disrupted and it can’t identify differences between foreign and body’s own cell.
Somatic hypermutation is a process using which different immunoglobulin molecules are formed through the combination of different gene variants. Millions of gene variants related to various pathogens and infections are there in our genome so predisposition studies to autoimmunity are quite difficult to establish.
One can’t say if someone will have autoimmunity or not! Studying just genes and mutations.
Conclusively, we can say, it is restricted, I mean, we can’t surely say whether someone will get a disease or not! Some of the limitations are explained here.
Limitations of genetic predisposition:
Polygenic nature:
Genetic susceptibility studies are designed to study mostly polygenetic disorders, in which many genes are associated. Studying multigenic disease is a time-consuming, costlier and manly process.
Lack of knowledge on the genome:
Scientists have sequenced the whole human genome. 23,000 different genes and non-coding regions are now known to us, however, many areas of the genome still remain unexplored.
Coding and non-coding DNA sequences react differently always and vary between individuals, species and persons. And therefore difficult to study at the population level.
The complexity of the genome:
Our genome is a complex thing. It’s made up of DNA, tightly wrapped using the protein on chromosomes. In addition to this, coding sequences (genes) need non-coding DNA too for expression.
The Complexity of the genome makes things harder to study and understand.
Complex pathways of chronic diseases:
Chronic diseases are complex in nature, many pathways and genes and gene sets are part of the process. The complex and complicated nature of the chronic disease makes it hard to study increases difficulty to many folds.
Lack of fully automated genome testing techniques:
Though many robust and fast genome sequencing platforms are available in recent times but aren’t enough to study genetic predisposition at the commercial level. Lack of fully automated technology increases cost and time.
Detection of genetic predisposition:
Common PCR and real-time setup aren’t enough to study as they can study only several genes or gene variants. State-of-the-art techniques such as whole-genome sequencing, whole-exome sequencing, whole-genome SNP array and genome microarray are the methods commonly employed in genetic predisposition and related studies.
Whole-genome sequencing:
A common sequencer like the sanger sequencer can sequence a few kbp DNA sequences which aren’t enough to study the genome. To investigate the whole-genome, next-generation sequencing and pyrosequencing techniques are practiced.
Those studies the entire DNA of a person’s genome; coding and non-coding both and finds variants; known and unknown. Meaning it can study many genes and variants at a time and henceforth used in genetic predisposition studies. You can read this article to learn more: 3 of the best whole-genome sequencing techniques.
Whole-exome sequencing:
The present technique is slightly different than the WGS. It can only sequence coding regions- exons of the gene. Variants that are present in the coding gene sequences are only sequenced.
Coding only exons are advantageous, at the same time disadvantageous too. It can reduce the cost and time of study but can’t produce huge data. Read this article to learn more: What is whole-exome sequencing.
Whole-genome SNP array:
SNPs are single nucleotide polymorphism or single-base changes that occur in our genome. Some SNPs are pathogenic while majorities are not. SNPs are the most popular marker in studying genetic predisposition.
SNPs profiles are commonly compared to produce data related to chronic disease and are used in investigating genetic susceptibility. If you are willing to learn more on SNPs, you can read this article: What is SNP?
DNA microarray:
DNA microarray is a hybridization-based genomic analysis technique, employed to investigate common variants present in the genome. On the downside, it can’t produce new data or study new variations.
However, it can study gene expression associated with a particular variant. Thousands or millions of known variants can be studied. Microarray is yet another best tool for genetic susceptibility study. Read this article to learn more: What is DNA microarray?
How does it help?
Now after reading all this technical stuff, you may wonder, is it really help us! If yes, how it can.
First, keep in mind that genetic predisposition shows only the probability of getting a chronic condition or disease. So using the data one may take preventive and management steps early in their life.
For example, if your risk of getting breast cancer is 35%, you can manage things. You can test timely to know the status. You can stay away from cancer-causing agents etc and take the doctor’s advice.
Meaning it indeed helps in saving your life. It can tell you;
- How you are susceptible to some disease?
- What is the probability of getting some kind of chronic disease?
- What disease is running in your family and your chances of getting it?
- What is the percentage you may develop some kind of cancer or autoimmune disease?
- That’s how it helps us, but unfortunately, as we said, not guaranteed.
Wrapping up:
While studying the present topic, keep two things in mind; genetic predisposition is just an assumption or probability of getting a disease and does not assure. It is useless to a person and put an extra economical burden, some group of scientists believes.
I personally agreed too, because even after paying $1000 to $2000, it will only tell us the probability, isn’t it worth it! Do comment and let me know.
But on the positive side, robust techniques like DNA sequencing and microarray help in many ways starting from disease diagnosis to manage prevention. It’s yet a dream to use genetic predisposition to study polygenic disease and is a long way off too.
Sources:
- Hamdi, Y., Boujemaa, M., Ben Rekaya, M. et al. Family specific genetic predisposition to breast cancer: results from Tunisian whole-exome sequenced breast cancer cases. J Transl Med 16, 158 (2018).
- Cancer predisposition by Peter Nelson Lab.
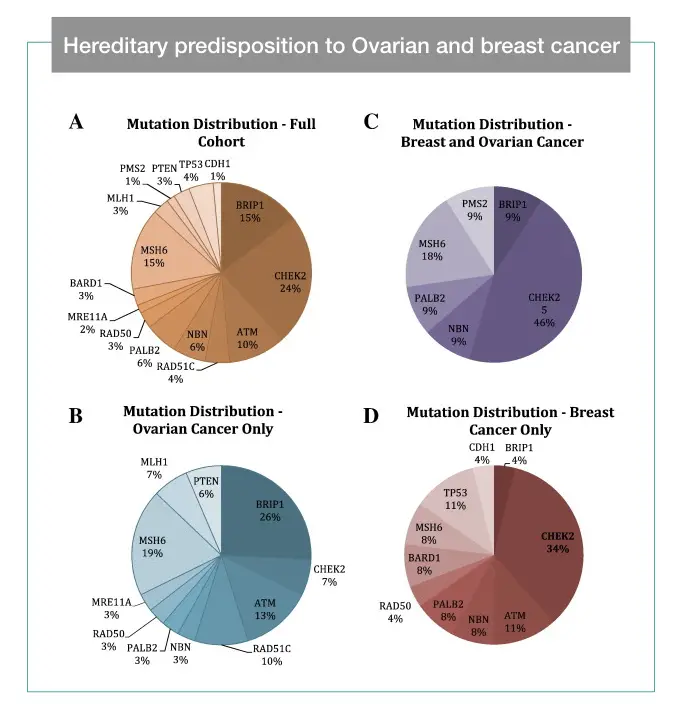
- Hereditary predisposition to ovarian cancer, looking beyond BRCA1/2- Lindsey EM et al.